1) Endosafe - PTS (Portable Test System)
|
|
|
- José Mangueira Vilalobos
- 8 Há anos
- Visualizações:
Transcrição
1
2
3
4
5 1) Endosafe - PTS (Portable Test System) 1.1. Descrição detalhada do produto médico, incluindo os fundamentos de seu funcionamento e sua ação, seu conteúdo ou composição, quando aplicável, assim como relação dos acessórios destinados a integrar o produto: Descrição do produto: O Endosafe - PTS (Portable Test System) é um sistema de teste rápido que é composto por um cartucho de teste de endotoxina juntamente com um espectrofotômetro portátil. O cartucho de teste utiliza reagentes de Limulus Amebocyte Lysate (LAL), sendo que este reagente possui registro na agência reguladora norte-americana - Food and Drug Administration (FDA). O Endosafe - PTS fornece resultados quantitativos do teste de LAL em aproximadamente 15 minutos. O Endosafe - PTS foi projetado para acelerar uma ampla gama de metodologias de testes de Controle de Qualidade. O teste de LAL é utilizado para detectar e/ou quantificar endotoxinas de origem bacteriana. O Endosafe - PTS é uma alternativa aos métodos de coloração de Gram convencionais para identificação dos micro-organismos em aproximadamente 3 a 7 minutos. Os cartuchos são pré-carregados com todos os reagentes necessários para realizar um ensaio de LAL, eliminando a preparação de diversos reagentes e reduzindo a possibilidade de erro técnico, uma vez que não há necessidade da elaboração de uma curva padrão com controle positivo e negativo. No entanto, recomenda-se que, ao realizar as diluições da amostra, o diluente deve ser testado para endotoxinas antes da sua utilização. Renata Ros Consultoria e Assessoria Empresarial Ltda. Brasília/DF: SRTVS QUADRA D 701 BLOCO O SALA 392 Centro Multiempresarial (61) Porto Alegre/RS: Rua Dr. Florêncio Ygartua, 271/ (51) [email protected] - 3
6 Figura 1: Visão geral do Endosafe - Portable Test System Fundamentos de seu funcionamento e sua ação: O Endosafe - PTS (Portable Test System) utiliza metodologia LAL cromogênico cinético que mede a intensidade da cor que está diretamente relacionada com a concentração de endotoxinas em uma amostra. Cada cartucho contém quantidades precisas de reagentes LAL, substrato cromogênico e endotoxina padrão controle (CSE). Os cartuchos são produzidos de acordo com rígidos procedimentos operacionais padrão promovendo a precisão do teste, consistência e estabilidade do produto. Para realizar o teste, o utilizador simplesmente pipeta 25 µl de uma amostra em cada uma das quatro amostras de reservatórios de cartucho. O leitor extrai e mistura a amostra com o reagente LAL em dois canais (os canais de amostra), e com o reagente de LAL e o controle positivo do produto nos outros dois canais (os canais de pico). A amostra é incubada e depois combinada com o substrato cromogênico. Após a mistura, a densidade óptica dos poços é medida e analisada contra uma curva padrão internamente arquivada. Renata Ros Consultoria e Assessoria Empresarial Ltda. Brasília/DF: SRTVS QUADRA D 701 BLOCO O SALA 392 Centro Multiempresarial (61) Porto Alegre/RS: Rua Dr. Florêncio Ygartua, 271/ (51) [email protected] - 4
7 Nesta metodologia, o cartucho do Endosafe - PTS (Portable Test System) realiza automaticamente uma duplicata da amostra e uma duplicata do teste LAL com controle positivo, de acordo com o preconizado com a harmonização da Farmacopeia USP para Teste de Endotoxinas Bacterianas. Conteúdo ou composição: Todos os componentes necessários para o ensaio de amostras com o Endosafe - PTS estão listados abaixo: Produto Descrição Catálogo Quantidade Endosafe PTS. Leitor e software integrado. Fonte de energia. Cabo do adaptador (1). Sistema de Teste Portátil PTS100 1 Pipetador Mini (1). Guia do usuário. Endosafe - PTS Licenciado. endotoxina Cartridge. (10 cartuchos por pacote). 0,1 EU/mL sensibilidade 0,05 EU/mL sensibilidade 0,01 EU/mL sensibilidade 0,005 EU/mL sensibilidade PTS201F PTS2005F PTS2001F PTS20005F 10 por embalagem Endosafe- PTS Unlicensed endotoxina Cartridge (10 cartuchos por pacote) 0,1 EU/mL sensibilidade 0,05 EU/mL sensibilidade 0,01 EU/mL sensibilidade PTS201 PTS2005 PTS por embalagem Renata Ros Consultoria e Assessoria Empresarial Ltda. Brasília/DF: SRTVS QUADRA D 701 BLOCO O SALA 392 Centro Multiempresarial (61) Porto Alegre/RS: Rua Dr. Florêncio Ygartua, 271/ (51) [email protected] - 5
8 PTS Endosafe- Inibição / Aperfeiçoamento Cartridge (10 cartuchos por pacote) Inibição/Aprimoramento tela PTS por embalagem PTS Endosafe- Inibição / Aprimoramento Cartridge (10 cartuchos por pacote) Inibição / Aprimoramento tela PTS por embalagem Endosafe- PTS rápida Micro métodos de cartucho (10 cartuchos por pacote) Gram ID LRMMGI por embalagem Endosafe- PTS Glucan Cartridge (10 cartuchos por pacote) (1,3) β-d-glucan RMMGS por embalagem Relação dos acessórios destinados a integrar o produto: Produto Descrição Código Cabo para PC AC adaptador Impressora Seiko DPU- 414 Cabo Moldado para PTS Reader para PC. Adaptador AC para o PTS Leitor. Alimentados por bateria, recarregável, unidade PTS108 PTS109 PTS300 Renata Ros Consultoria e Assessoria Empresarial Ltda. Brasília/DF: SRTVS QUADRA D 701 BLOCO O SALA 392 Centro Multiempresarial (61) Porto Alegre/RS: Rua Dr. Florêncio Ygartua, 271/ (51) [email protected] - 6
9 Papel de impressora Seiko Cabo de impressora Seiko Epson TM-U220D de impressora Cabo da impressora Epson Fita para impressora Epson Papel Epson PTS Endosafe - mini-pipetador Caso leitor de proteção Ponteiras de pipeta Eppendorf térmica. Rolos de papel térmico para Seiko. DPU-414 de impressora. Moldado cabo para PTS Reader para Seiko DPU- 414 de impressora. Unidade não-térmica alimentado AC. Moldado cabo para Epson TM-U220D de impressora. Fita para Epson TM-220D de impressora. Rolos de papel de impressora para Epson TM-U220D. 25 µl unidade descartável com + 5% de precisão e + 3% de precisão em curso completo. Plástico sólido, com alça para instrumentos e acessórios 200 µl, embalados individualmente, validado para 0,005 EU/mL PTS301 PTS302 PTS310 PTS311 PTS312 PTS313 PTS400 PTS600 D Indicação, finalidade ou uso a que se destina o produto médico, segundo indicado pelo fabricante: O Endosafe - PTS (Portable Test System) é um sistema de teste rápido que é composto por um cartucho de teste de endotoxina juntamente com um Renata Ros Consultoria e Assessoria Empresarial Ltda. Brasília/DF: SRTVS QUADRA D 701 BLOCO O SALA 392 Centro Multiempresarial (61) Porto Alegre/RS: Rua Dr. Florêncio Ygartua, 271/ (51) [email protected] - 7
10 espectrofotômetro portátil. O teste de LAL é utilizado para detectar e/ou quantificar endotoxinas de origem bacteriana. O Endosafe - PTS fornece resultados quantitativos do teste de LAL em aproximadamente 15 minutos. O Endosafe - PTS foi projetado para acelerar uma ampla gama de metodologias de testes de Controle de Qualidade Precauções, restrições, advertências, cuidados especiais e esclarecimentos sobre o uso do produto médico, assim como seu armazenamento e transporte: NÃO utilizar um adaptador AC diferente do que foi entregue com seu sistema. Apenas ligue o adaptador a uma tomada que está ligado à terra. Usando um adaptador diferente pode causar incêndio que conduz a lesões graves. NÃO coloque sistema PTS ou carga da bateria em torno de quaisquer líquidos ou a utilização num local onde ele poderia se molhar ou salpicar. Os líquidos podem causar incêndio ou choque elétrico. NÃO abra o instrumento e mexa nos componentes internos. Ele não contém peças que podem ser mantidos, reparados ou substituídos pelo usuário. Isso pode causar incêndio ou choque elétrico. NÃO permita que líquidos sejam derramados para dentro da câmara do cartucho. Isso pode danificar os componentes internos e pode provocar um incêndio ou choque elétrico. NÃO use objetos pontiagudos para operar o teclado de seu sistema. NÃO desligue o cabo de alimentação CA ou o cabo PC puxando fio/cabo diretamente. Isso pode danificar cabo e causar ferimentos. Renata Ros Consultoria e Assessoria Empresarial Ltda. Brasília/DF: SRTVS QUADRA D 701 BLOCO O SALA 392 Centro Multiempresarial (61) Porto Alegre/RS: Rua Dr. Florêncio Ygartua, 271/ (51) [email protected] - 8
11 Lidar com o adaptador AC e Porta Serial diretamente com o conector para desconectar. Leia este Manual do Usuário antes de utilizar o Sistema Endosafe - PTS. A Compreensão o uso adequado de operação é fundamental para a segurança do usuário, bem como para proteção do Sistema Endosafe - PTS. Armazenamento e Transporte: O produto pode ser transportado e armazenado em temperaturas na faixa de 15 a 27ºC, e umidade de 5 a 95% (sem condensação) Formas de apresentação do produto médico: As formas de apresentação do produto Endosafe - PTS (Portable Test System) estão descritas acima no item Conteúdo e Composição. Imagens gráficas do produto (fotos, desenhos técnicos, etc.): Figura 2: Endosafe - Portable Test System. Renata Ros Consultoria e Assessoria Empresarial Ltda. Brasília/DF: SRTVS QUADRA D 701 BLOCO O SALA 392 Centro Multiempresarial (61) Porto Alegre/RS: Rua Dr. Florêncio Ygartua, 271/ (51) [email protected] - 9
12 Figura 3: Visão geral do Endosafe - Portable Test System. Figura 4: Endosafe - Portable Test System e Cartuchos Cartridges. Renata Ros Consultoria e Assessoria Empresarial Ltda. Brasília/DF: SRTVS QUADRA D 701 BLOCO O SALA 392 Centro Multiempresarial (61) Porto Alegre/RS: Rua Dr. Florêncio Ygartua, 271/ (51) [email protected]
13 Figura 5: Endosafe - Portable Test System com cartuchos cartridges, impressora, pipetas e ponteiras. Figura 6: Cartuchos cartridges para utilização no Endosafe - Portable Test System. Renata Ros Consultoria e Assessoria Empresarial Ltda. Brasília/DF: SRTVS QUADRA D 701 BLOCO O SALA 392 Centro Multiempresarial (61) Porto Alegre/RS: Rua Dr. Florêncio Ygartua, 271/ (51) [email protected]
14 2) Endosafe MCS (Multi-Cartridge System) 1.1. Descrição detalhada do produto médico, incluindo os fundamentos de seu funcionamento e sua ação, seu conteúdo ou composição, quando aplicável, assim como relação dos acessórios destinados a integrar o produto: Descrição do produto: O Endosafe - MCS (Multi Cartridge System) é um sistema de detecção de endotoxina multi-cartucho que utiliza reagentes de Limulus Amebocyte Lysate (LAL), sendo que este reagente possui registro na agência reguladora norte-americana - Food and Drug Administration (FDA) - e múltiplos espectrofotômetros para fornecer resultados de teste de endotoxinas para até 5 amostras em cerca de 15 minutos. Os cartuchos Cartridge são os mesmos utilizados na versão portátil (PTS). Os resultados quantitativos dos testes de LAL são capturados via o software de detecção de endotoxina EndoScan-V, produzido pelo fabricante Charles River. O Endosafe - MCS é composto por 5 espectrofotômetros individuais construídos em uma unidade com um conector USB único que liga a um computador desktop. O MCS utiliza a mesma metodologia já descrita para o produto Endosafe - PTS. Figura 1: Visão geral do Endosafe MCS (Multi-Cartridge System) Renata Ros Consultoria e Assessoria Empresarial Ltda. Brasília/DF: SRTVS QUADRA D 701 BLOCO O SALA 392 Centro Multiempresarial (61) Porto Alegre/RS: Rua Dr. Florêncio Ygartua, 271/ (51) [email protected]
15 Fundamentos de seu funcionamento e sua ação: O Endosafe - MCS (Multi Cartridge System) utiliza metodologia LAL cromogênico cinético que mede a intensidade da cor que está diretamente relacionada com a concentração de endotoxinas em uma amostra. Cada cartucho contém quantidades precisas de reagentes LAL, substrato cromogênico e endotoxina padrão controle (CSE). Os cartuchos são produzidos de acordo com rígidos procedimentos operacionais padrão promovendo a precisão do teste, consistência e estabilidade do produto. Para realizar o teste, o utilizador simplesmente pipeta 25 µl de uma amostra em cada uma das quatro amostras de reservatórios de cartucho. O leitor extrai e mistura a amostra com o reagente LAL em dois canais (os canais de amostra), e com o reagente de LAL e o controle positivo do produto nos outros dois canais (os canais de pico). A amostra é incubada e depois combinada com o substrato cromogênico. Após a mistura, a densidade óptica dos poços é medida e analisada contra uma curva padrão internamente arquivada. Conteúdo ou composição: Todos os componentes necessários para o ensaio de amostras com o Endosafe - MCS estão listados abaixo: Produto Descrição Catálogo Quantidade Kit Endosafe MCS. Leitor e software integrado. Fonte de energia integrado. Sistema Multi-Cartucho MCS100 1 Cabo de força. Renata Ros Consultoria e Assessoria Empresarial Ltda. Brasília/DF: SRTVS QUADRA D 701 BLOCO O SALA 392 Centro Multiempresarial (61) Porto Alegre/RS: Rua Dr. Florêncio Ygartua, 271/ (51) [email protected]
16 Guia do usuário. Endosafe Pacote de Cartuchos Licenciados. (10 cartuchos por pacote). 0,1 EU/mL sensibilidade 0,05 EU/mL sensibilidade 0,01 EU/mL sensibilidade 0,005 EU/mL sensibilidade PTS201F PTS2005F PTS2001F PTS20005F 1 por embalagem Endosafe - Cartuchos Não licenciados (10 cartuchos por pacote) 0,1 EU/mL sensibilidade 0,05 EU/mL sensibilidade 0,01 EU/mL sensibilidade PTS201 PTS2005 PTS por embalagem Endosafe - Multi - Embalagem Cartridge (25 cartuchos por pacote) 5 to 0.05 EU/mL sensibilidade 1 to 0.01 EU/mL sensibilidade 0.5 to EU/mL sensibilidade PTS5505F PTS5501F PTS55005F 5 Cartuchos e 5 embalagens Endosafe - Multi - Embalagem Cartridge 50 cartuchos por pacote) 5 to 0.05 EU/mL sensibilidade 1 to 0.01 EU/mL sensibilidade PTS1105F PTS1101F 5 Cartuchos e 10 embalagens Endosafe - Multi - Embalagem Cartridge 100 cartuchos por pacote) 5 to 0.05 EU/mL sensibilidade 1 to 0.01 EU/mL sensibilidade PTS1105F PTS1101F 10 Cartuchos e 10 embalagens Endosafe- PTS Screening Inibição / Aprimoramento Cartridge Screening Inibição / Aprimoramento PTS220 1 por embalagem (10 cartuchos por pacote) Endosafe - PTS RMM Gram ID Cartridge Endosafe - PTS RMM Gram ID LRMMGI100 1 por embalagem (1,3) β-d-glucan RMMGS por Renata Ros Consultoria e Assessoria Empresarial Ltda. Brasília/DF: SRTVS QUADRA D 701 BLOCO O SALA 392 Centro Multiempresarial (61) Porto Alegre/RS: Rua Dr. Florêncio Ygartua, 271/ (51) [email protected]
17 Glucan Cartridge embalagem (10 cartuchos por pacote) Relação dos acessórios destinados a integrar o produto: Produto Descrição Código MCS Endosafe - mini-pipetador Ponteiras de pipeta Eppendorf 25 µl unidade descartável 200 µl, embalados individualmente, validado para 0,005 EU/mL PTS400 D Indicação, finalidade ou uso a que se destina o produto médico, segundo indicado pelo fabricante: O Endosafe - MCS (Multi Cartridge System) é um sistema de detecção de endotoxina multi-cartucho que utiliza reagentes de Limulus Amebocyte Lysate (LAL) e múltiplos espectrofotômetros para fornecer resultados de teste de endotoxinas bacterianas para até 5 amostras em cerca de 15 minutos. O Endosafe - MCS pode testar 5 cartuchos simultaneamente, ou qualquer combinação de 1 a 5. As amostras são executadas de forma independente, permitindo acesso aleatório, sendo que diversas unidades do Endosafe - MCS podem ser ligados a um único computador para um elevado rendimento de amostras com resultados em tempo real. Com o Endosafe - MCS, a comunicação dos dados é simples. Na conclusão do teste, os critérios de medição de endotoxinas e de aceitação do ensaio são calculados pelo software EndoScan-V e são exibidas na tela do computador. O instrumento pode ser utilizado para detectar níveis de endotoxinas elevados como 10 UE/mL e baixas como 0,005 EU/mL. Os relatórios detalhados podem ser gerados a partir do Endosafe - MCS através Renata Ros Consultoria e Assessoria Empresarial Ltda. Brasília/DF: SRTVS QUADRA D 701 BLOCO O SALA 392 Centro Multiempresarial (61) Porto Alegre/RS: Rua Dr. Florêncio Ygartua, 271/ (51) [email protected]
18 do software EndoScan-V, sendo exportados fazer o programa Excel, KIMS e outras bases de dados centralizadas. O Endosafe - MCS foi projetado para acelerar uma ampla gama de metodologias de testes de Controle de Qualidade Precauções, restrições, advertências, cuidados especiais e esclarecimentos sobre o uso do produto médico, assim como seu armazenamento e transporte: NÃO utilizar um adaptador AC diferente do que foi entregue com seu sistema. Apenas ligue o adaptador a uma tomada que está ligado à terra. Usando um adaptador diferente pode causar incêndio que conduz a lesões graves. NÃO coloque sistema PTS ou carga da bateria em torno de quaisquer líquidos ou a utilização num local onde ele poderia se molhar ou salpicar. Os líquidos podem causar incêndio ou choque elétrico. NÃO abra o instrumento e mexa nos componentes internos. Ele não contém peças que podem ser mantidos, reparados ou substituídos pelo usuário. Isso pode causar incêndio ou choque elétrico. NÃO permita que líquidos sejam derramados para dentro da câmara do cartucho. Isso pode danificar os componentes internos e pode provocar um incêndio ou choque elétrico. NÃO use objetos pontiagudos para operar o teclado de seu sistema. NÃO desligue o cabo de alimentação CA ou o cabo PC puxando fio/cabo diretamente. Isso pode danificar cabo e causar ferimentos. Lidar com o adaptador AC e Porta Serial diretamente com o conector para desconectar. Renata Ros Consultoria e Assessoria Empresarial Ltda. Brasília/DF: SRTVS QUADRA D 701 BLOCO O SALA 392 Centro Multiempresarial (61) Porto Alegre/RS: Rua Dr. Florêncio Ygartua, 271/ (51) [email protected]
19 Leia este Manual do Usuário antes de utilizar o Sistema Endosafe - MCS. A Compreensão o uso adequado de operação é fundamental para a segurança do usuário, bem como para proteção do Sistema Endosafe - MCS. Armazenamento e Transporte: O produto pode ser transportado e armazenado em temperaturas na faixa de 15 a 27ºC, e umidade de 5 a 80% (sem condensação). Uso somente em ambiente interno (indoor) Formas de apresentação do produto médico: As formas de apresentação do produto Endosafe - MCS (Multi Cartridge System) estão descritas acima no item Conteúdo e Composição. Imagens gráficas do produto (fotos, desenhos técnicos, etc.): Figura 2: Vista frontal do Sistema Endosafe - MCS (Multi Cartridge System) Renata Ros Consultoria e Assessoria Empresarial Ltda. Brasília/DF: SRTVS QUADRA D 701 BLOCO O SALA 392 Centro Multiempresarial (61) Porto Alegre/RS: Rua Dr. Florêncio Ygartua, 271/ (51) [email protected]
20 Figura 3: Vista lateral do Sistema Endosafe - MCS (Multi Cartridge System) Figura 4: Vista traseira esquerda do Sistema Endosafe - MCS (Multi Cartridge System) Renata Ros Consultoria e Assessoria Empresarial Ltda. Brasília/DF: SRTVS QUADRA D 701 BLOCO O SALA 392 Centro Multiempresarial (61) Porto Alegre/RS: Rua Dr. Florêncio Ygartua, 271/ (51) [email protected]
21 Figura 5: Vista traseira direita do Sistema Endosafe - MCS (Multi Cartridge System) Figura 6: Cartuchos cartridge do Sistema Endosafe - MCS (Multi Cartridge System) Renata Ros Consultoria e Assessoria Empresarial Ltda. Brasília/DF: SRTVS QUADRA D 701 BLOCO O SALA 392 Centro Multiempresarial (61) Porto Alegre/RS: Rua Dr. Florêncio Ygartua, 271/ (51) [email protected]
22 3) Endosafe Nexus 1.1. Descrição detalhada do produto médico, incluindo os fundamentos de seu funcionamento e sua ação, seu conteúdo ou composição, quando aplicável, assim como relação dos acessórios destinados a integrar o produto: Descrição do produto: O Endosafe Nexus é o primeiro sistema robótico totalmente automatizado concebido especificamente para o teste de endotoxina no laboratório central de Controle de Qualidade. É Ideal para testes da qualidade da água de grande volume ou amostras que necessitam de diluições. O sistema utiliza a comprovada tecnologia inovadora de cartucho com reagentes de LAL (Limulus Amebocyte Lysate,) e um simples software de gerenciamento de dados para testar 48 a 60 amostras com preparação e supervisão mínima. Figura 1: Visão geral do Equipamento Endosafe Nexus O sistema oferece a oportunidade única para reduzir o tempo técnico e maximizar o espaço de laboratório, enquanto diminui a variabilidade do teste e a necessidade de investigações subsequentes. O Endosafe Nexus fornece o Renata Ros Consultoria e Assessoria Empresarial Ltda. Brasília/DF: SRTVS QUADRA D 701 BLOCO O SALA 392 Centro Multiempresarial (61) Porto Alegre/RS: Rua Dr. Florêncio Ygartua, 271/ (51) [email protected]
23 teste de LAL de alto rendimento, livre de erros, em uma baixa manutenção, em um pacote de alta precisão. Fundamentos de seu funcionamento e sua ação: O sistema robótico Endosafe Nexus utiliza métodologia LAL cromogênico cinético que mede a intensidade da cor que está diretamente relacionada com a concentração de endotoxinas em uma amostra. Cada cartucho contém quantidades precisas de reagentes LAL, substrato cromogênico e endotoxina padrão controle (CSE). Os cartuchos são produzidos de acordo com rígidos procedimentos operacionais padrão promovendo a precisão do teste, consistência e estabilidade do produto, seguindo as recomendações farmacopéicas, preenchendo todos os critérios específicos para técnicas fotométricas. O sistema oferece a oportunidade única para reduzir o tempo técnico e maximizar o espaço de laboratório, enquanto diminui a variabilidade do teste e a necessidade de investigações subsequentes. Conteúdo ou composição: 1 Sistema Robótico Endosafe Nexus. Figura 2: Configuração da plataforma do Sistema Robótico Endosafe Nexus Renata Ros Consultoria e Assessoria Empresarial Ltda. Brasília/DF: SRTVS QUADRA D 701 BLOCO O SALA 392 Centro Multiempresarial (61) Porto Alegre/RS: Rua Dr. Florêncio Ygartua, 271/ (51) [email protected]
24 Relação dos acessórios destinados a integrar o produto: Não se aplica Indicação, finalidade ou uso a que se destina o produto médico, segundo indicado pelo fabricante: O Endosafe Nexus é o primeiro sistema robótico totalmente automatizado concebido especificamente para o teste de endotoxina no laboratório central de Controle de Qualidade. É Ideal para testes da qualidade da água de grande volume ou amostras que necessitam de diluições. O sistema utiliza a comprovada tecnologia inovadora de cartucho com reagentes de LAL (Limulus Amebocyte Lysate) e um simples software de gerenciamento de dados para testar 48 a 60 amostras com preparação e supervisão mínima Precauções, restrições, advertências, cuidados especiais e esclarecimentos sobre o uso do produto médico, assim como seu armazenamento e transporte: Não se aplica Formas de apresentação do produto médico: 1 Sistema Robótico Endosafe Nexus. Sistema robótico totalmente automatizado concebido especificamente para o teste de endotoxina por LAL (Limulus Amebocyte Lysate). Renata Ros Consultoria e Assessoria Empresarial Ltda. Brasília/DF: SRTVS QUADRA D 701 BLOCO O SALA 392 Centro Multiempresarial (61) Porto Alegre/RS: Rua Dr. Florêncio Ygartua, 271/ (51) [email protected]
25 Imagens gráficas do produto (fotos, desenhos técnicos, etc.): Figura 3: Sistema Robótico Endosafe Nexus Renata Ros Consultoria e Assessoria Empresarial Ltda. Brasília/DF: SRTVS QUADRA D 701 BLOCO O SALA 392 Centro Multiempresarial (61) Porto Alegre/RS: Rua Dr. Florêncio Ygartua, 271/ (51) [email protected]
26 4) Endosafe Microtrend (Software) 1.1. Descrição detalhada do produto médico, incluindo os fundamentos de seu funcionamento e sua ação, seu conteúdo ou composição, quando aplicável, assim como relação dos acessórios destinados a integrar o produto: Descrição do produto: Endosafe Microtrend é um software com um banco de dados específico para o teste de LAL (Limulus Amebocyte Lysate). Ele trabalha em conjunto com outro software intitulado EndoScan-V produzido pelo mesmo Fabricante, para organizar e analisar os dados de testes de LAL. O software Endosafe Microtrend pode ser adaptado para atender a uma série de objetivos, incluindo a apresentação dos resultados ilustrados através de uma rede, facilitando a revisão dos dados disponíveis. Fundamentos de seu funcionamento e sua ação: Não se aplica. Conteúdo ou composição: 1 Software Endosafe Microtrend. Relação dos acessórios destinados a integrar o produto: Não se aplica. Renata Ros Consultoria e Assessoria Empresarial Ltda. Brasília/DF: SRTVS QUADRA D 701 BLOCO O SALA 392 Centro Multiempresarial (61) Porto Alegre/RS: Rua Dr. Florêncio Ygartua, 271/ (51) [email protected]
27 1.2. Indicação, finalidade ou uso a que se destina o produto médico, segundo indicado pelo fabricante: Endosafe Microtrend é um software com um banco de dados específico para o teste de LAL (Limulus Amebocyte Lysate). O referido software permite compilar, organizar e analisar dados históricos de testes LAL, sem a necessidade da inserção dos dados em uma planilha para posterior análise, permitindo a visualização de dados ao longo de um período de tempo. O software Endosafe Microtrend pode ser adaptado para atender a uma série de objetivos, incluindo a apresentação dos resultados ilustrados através de uma rede, facilitando a revisão dos dados disponíveis Precauções, restrições, advertências, cuidados especiais e esclarecimentos sobre o uso do produto médico, assim como seu armazenamento e transporte: Não se aplica Formas de apresentação do produto médico: 1 Software Endosafe Microtrend. Imagens gráficas do produto (fotos, desenhos técnicos, etc.): Não se aplica. As imagens das capturas de telas do referido software em execução podem ser verificadas na instrução de uso do produto enviada em anexo. Renata Ros Consultoria e Assessoria Empresarial Ltda. Brasília/DF: SRTVS QUADRA D 701 BLOCO O SALA 392 Centro Multiempresarial (61) Porto Alegre/RS: Rua Dr. Florêncio Ygartua, 271/ (51) [email protected]
28 5) EndoScan-V version 5 (Software) 1.1. Descrição detalhada do produto médico, incluindo os fundamentos de seu funcionamento e sua ação, seu conteúdo ou composição, quando aplicável, assim como relação dos acessórios destinados a integrar o produto: Descrição do produto: EndoScan-V é um software de medição de endotoxina compatível com uma variedade de leitores de placas. É validado para estar de acordo com os requisitos exigidos pelo FDA na realização dos cálculos necessários para liberação de lote de produto. O programa foi projetado com capacidades de configuração flexíveis de trabalho em rede e opções de desempenho para facilitar ganhos de eficiência operacional. Fundamentos de seu funcionamento e sua ação: Não se aplica. Conteúdo ou composição: 1 Software EndoScan-V version 5. Relação dos acessórios destinados a integrar o produto: Não se aplica. Renata Ros Consultoria e Assessoria Empresarial Ltda. Brasília/DF: SRTVS QUADRA D 701 BLOCO O SALA 392 Centro Multiempresarial (61) Porto Alegre/RS: Rua Dr. Florêncio Ygartua, 271/ (51) [email protected]
29 1.2. Indicação, finalidade ou uso a que se destina o produto médico, segundo indicado pelo fabricante: EndoScan-V é um software de medição para o teste de LAL para a detecção de endotoxina bacterianas Precauções, restrições, advertências, cuidados especiais e esclarecimentos sobre o uso do produto médico, assim como seu armazenamento e transporte: Não se aplica Formas de apresentação do produto médico: 1 Software EndoScan-V versão 5. Imagens gráficas do produto (fotos, desenhos técnicos, etc.): Não se aplica. As imagens das capturas de telas do referido software em execução podem ser verificadas na instrução de uso do produto enviada em anexo. Renata Ros Consultoria e Assessoria Empresarial Ltda. Brasília/DF: SRTVS QUADRA D 701 BLOCO O SALA 392 Centro Multiempresarial (61) Porto Alegre/RS: Rua Dr. Florêncio Ygartua, 271/ (51) [email protected]
30 Copia da pergunta Protocolo Fale Conosco - ANVISA
31 Copia da Pergunta realizada no dia 12/03/2015: Protocolo do Fale Conosco da ANVISA: ATENÇÃO GQUIP - GGTPS: Solicito esclarecimento para a GQUIP da GGTPS, se Equipamentos para Detecção de Endotoxinas são passíveis de Registro na ANVISA ou não, uma vez que não são para detecção em saúde (In Vitro). Obrigado. Seu número de Protocolo é:
32
33 Copia da resposta Protocolo Fale Conosco - ANVISA
34 Zarafa WebAccess - Carlos Wayhs - Oriens Consultoria 1 de 1 07/08/ :50 Carlos Wayhs - Oriens Consultoria De: Central de Atendimento ao Público - Anvisa <[email protected]> Enviado: Qui 12/03/ :35 Para: Carlos Wayhs - Oriens Consultoria <[email protected]> Assunto: Anvisa - Resposta ao protocolo Modificado: Sex 13/03/ :34 Prezado (a) Senhor (a), Em atenção a sua solicitação, informamos que de acordo com a Resolução RDC nº 185/01, considera-se produto para saúde o material, artigo ou sistema de uso ou aplicação médica, odontológica ou laboratorial, destinado à prevenção, diagnóstico, tratamento, reabilitação ou anticoncepção; que não utiliza meio farmacológico, imunológico ou metabólico para realizar sua principal função em seres humanos, podendo, entretanto, ser auxiliado em suas funções por tais meios; e cujo funcionamento não depende de fonte de energia elétrica ou qualquer outra fonte de potência distinta da gerada pelo corpo humano ou gravidade e que funciona pela conversão desta energia. Dúvidas quanto ao enquadramento do produto No caso de dúvidas quanto ao enquadramento de produto, deverá ser protocolizada uma carta na Anvisa contendo: - as informações sobre o produto indicadas nos itens 1.1 a 1.4 do Relatório Técnico contido no Anexo III.C do regulamento técnico aprovado pela Resolução RDC nº 185/01; e - imagens gráficas do produto (fotos, desenhos técnicos, etc.). Nesse caso, não há necessidade de encaminhamento de amostras do produto para a ANVISA. Atenciosamente Anvisa atende Central de atendimento Agência Nacional de Vigilância Sanitária Siga a Anvisa: Este endereço eletrônico está habilitado apenas para enviar s. Caso deseje entrar em contato com a central, favor ligar no ou acessar o Fale conosco, disponível no portal da Anvisa (link: ( /faleconosco/faleconosco.asp). As ligações podem ser feitas de segunda a sexta - feira, das 7h30 às 19h30, exceto feriados.
35 Instruções de Uso do Produto 1: Endosafe PTS (Portable Test System)
36 The Endosafe -PTS Portable Test System USER S GUIDE Version 7 endotoxin and microbial detection 1023 Wappoo Road Suite 43-B, Charleston, SC Phone: Fax: [email protected]
37 Endosafe - Portable Test System User s Guide Table of Contents Safety Guidelines... 1 Endosafe - PTS System Overview Reader Specifications... 2 Cartridge Specifications... 3 Accessory Products... 4 Endosafe - PTS Service Programs Warranty with Purchase... 5 Annual Calibration Certification... 5 Extended Warranty Program... 5 On Demand Repairs... 6 Returned Goods Authorization Procedure... 6 Warranty Registration... 6 Operating the Reader Battery Information... 7 Keypad Operation... 8 ON/OFF... 8 MENU Key... 8 Example of Keypad Operation Menu Shortcuts for PTS Reader Keypad Cancel Key Reader Output Downloading Output to PC using the PTS Logger Software Printing with the DPU-414 Seiko Thermal Printer or the Epson TM-U220D Printer Using EndoScan-V with Endosafe PTS for data acquisition...14 Troubleshooting Copyright 2003 PIPTS101-10
38 Endosafe - Portable Test System User s Guide Appendixes Appendix A Performing Routine Endotoxin Test... A1 Endotoxin Test Results... A2 Retrieving Results Options... A3 Appendix B Performing Routine Gram ID Test... B1 Gram ID Test Results... B3 Retrieving Results Options... B4 Appendix C Performing Inhibition/Enhancement Test... C1 Inhibition/Enhancement Test Results... C3 Retrieving Results Options... C4 Appendix D Performing Routine Glucan Test... D1 Glucan Test Results... D2 Retrieving Results Options... D3 Appendix E Abbreviations Used... E1 Appendix F Decontamination Procedures... F1 Copyright 2003 PIPTS101-10
39 Endosafe - Portable Test System User s Guide SAFETY FIRST WARNING DO NOT use an AC power adapter other than that which has been delivered with your system. Only plug the adapter into a socket which is grounded. Using a different adapter may cause fire leading to serious injury. DO NOT place PTS system or charge battery around any liquids or use in a location where it could get wet or splashed. Liquids could cause fire or electric shock. CAUTION DO NOT open the instrument and tamper with the interior components. It does not contain any parts that can be maintained, repaired, or replaced by the user. Doing so may cause fire or electric shock. DO NOT allow liquids to spill into the cartridge chamber. Doing so may damage interior components and may cause fire or electric shock. DO NOT use sharply pointed objects to operate the keyboard of your system. DO NOT unplug AC power cord or PC cable by pulling cord/cable directly. Doing so may damage cord and cause injury. Handle the AC adapter and Serial Port Connector directly to unplug. Read this User s Guide carefully before using the Endosafe - PTS System. Understanding- the proper usage and operation is imperative for the safety of the User, as well as the protection of the Endosafe - PTS System. Copyright 2003 Page 1 PIPTS101-10
40 Endosafe - Portable Test System User s Guide Endosafe - PTS System Overview The Endosafe - PTS (Portable Test System) is a rapid, point-of-use test system which is comprised of a test cartridge along with a hand-held spectrophotometer. The Endosafe - PTS Endotoxin test utilizes existing FDA-licensed LAL formulations. The PTS provides quantitative LAL test results in approximately 15 minutes (see Appendix A for testing procedures and test results). The Endosafe - PTS Rapid Micro Methods are cartridges designed to accelerate a wide range of quality control test methodologies. New assays are currently in development and will be available as we adapt them to the PTS format. The Rapid Micro Methods Gram ID assay is an alternative to conventional gram stain methods for identification of microorganisms in approximately 3 to 7 minutes (see Appendix B for testing procedure and test results). All components required for testing samples with the PTS are listed below. Product Description Catalog # Quantity Endosafe - PTS kit Reader and integrated software Power supply Adapter cord (1) Mini pipettor (1) User s Guide Endosafe - Licensed PTS Endotoxin Cartridge (10 cartridges per package) Endosafe - Unlicensed PTS Endotoxin Cartridge (10 cartridges per package) Endosafe - PTS Inhibition/Enhancement Cartridge (10 cartridges per package) Endosafe - PTS Rapid Micro Methods Cartridge (10 cartridges per package) Portable Test System 0.1 EU/mL sensitivity 0.05 EU/mL sensitivity 0.01 EU/mL sensitivity EU/mL sensitivity 0.1 EU/mL sensitivity 0.05 EU/mL sensitivity 0.01 EU/mL sensitivity Inhibition/Enhancement Screen PTS100 PTS201F PTS2005F PTS2001F PTS20005F PTS201 PTS2005 PTS2001 PTS220 Each 10/pack 10/pack 10/pack Gram ID LRMMGI100 10/pack Endosafe - PTS Glucan Cartridge (10 cartridges per package) (1,3) -D-Glucan RMMGS /pack Note: Multi-cartridge packs are also available. Visit online at for additional product information..endosafe - PTS Reader Specifications Temperature control: 37º C + 1º C Data interface: RS232 serial port Printer support: Thermal printer and dot matrix compatible Data storage: Maximum of one hundred (100) stored test results. Copyright 2003 Page 2 PIPTS101-10
41 Endosafe - Portable Test System User s Guide Dimensions: 9.25 x x 2.50 corresponding 235 mm (D) x 128 mm (W) x 65 mm (H) Power supply unit: V, Hertz Power supply on board: 12 V DC, 5A Battery life: 6-8 hours of operation post-charge Warm-up time: Approximately 5-10 minutes from 20º C start Display: Two (2) lines of sixteen (16) characters each Environmental Conditions: Operating Temperature 15º to 27 º C Humidity 5% to 95% noncondensing Indoor and outdoor use Altitude - unlimited Weight: 3 lbs. corresponding 1100 Gram (without power supply unit) Endosafe - PTS Cartridge Specifications Reference package insert for storage requirements Endotoxin Sensitivity: EU/mL range, EU/mL range, EU/mL range, EU/mL range (see Appendix A) Rapid Micro Methods Gram ID: Archived parameters for Gram-negative, Grampositive and Yeast/Mold isolate identification (see Appendix B) Inhibition/Enhancement Screening (see Appendix C) Rapid Micro Methods Glucan (see Appendix D) Acceptable temperature: 37º C + 1º C Sample requirement: 25 μl sample per channel NOTE: Sample must be delivered to each sample port without contamination or cross contamination between samples. Copyright 2003 Page 3 PIPTS101-10
42 Endosafe - Portable Test System User s Guide Endosafe - PTS Accessory Products Product Description Code PC Cable Molded cable for PTS Reader to PTS108 PC AC Adaptor AC Adaptor for the PTS Reader PTS109 Seiko DPU-414 Printer Battery powered, rechargeable PTS300 thermal unit Seiko Printer Paper Thermal paper rolls for Seiko PTS301 DPU-414 Printer Seiko Printer Cable Molded cable for PTS Reader to PTS302 Seiko DPU-414 Printer Epson TM-U220D Printer AC powered non-thermal unit PTS310 Epson Printer Cable Molded cable for Epson TM-U220D Printer Epson Printer Ribbon Ribbon for Epson TM-U220D Printer Epson Printer Paper Paper rolls for Epson TM-U220D Printer Endosafe - PTS mini pipettor 25 μl disposable unit with + 5% accuracy and + 3% precision at full stroke Protective reader case Solid plastic, with handle for instruments and supplies Eppendorf * pipette tips 200 μl, individually wrapped, validated to EU/mL *Eppendorf is a registered trademark of Brinkmann PTS311 PTS312 PTS313 PTS400 PTS600 D100 Multiple patents pending. Copyright 2003 Page 4 PIPTS101-10
43 Endosafe - Portable Test System User s Guide Endosafe - PTS Service Programs WARRANTY with PURCHASE Charles River Laboratories warrants the Endosafe - PTS instrument will be delivered in a functional state and free from defect, and will meet stated specifications for a period of one (1) year. The warranty period will start on the date of shipment by Charles River Laboratories. During the warranty period, Charles River Laboratories will repair the instrument at no charge to the client for parts and labor. If repairs are needed, Charles River Laboratories will provide the client a loaner instrument during the period the instrument is being repaired. The client shall be responsible for all freight charges associated with the returned instrument and the loaner instrument. Opening the case or tampering with the interior components of the PTS Reader will invalidate warranty provisions. Additional service contracts are available as follows: Service Annual Calibration Certification Extended Warranty IQ,OQ,PQ On Demand Repairs Code PTS500 PTS501 PTS502 PTS503 ANNUAL CALIBRATION CERTIFICATION The PTS instrument factory calibration, which is certified at the time of initial shipment, is valid for one (1) year. Charles River Laboratories offers an annual re-calibration service. To request this service, the client must notify the Company sixty (60) days prior to the end of the initial calibration period to arrange for re-calibration service. EXTENDED WARRANTY PROGRAM The Extended Warranty Program is available to the client during the 1 st year from the date of purchase of the initial Warranty Period. This service program includes: A. A diagnostic and performance check, including replacement or repair of parts as necessary to return the instrument to original performance specifications. B. Loading software improvements/upgrades introduced to the PTS design since last inspection. C. A complete instrument cleaning operation. D. Certification for the extension of the Warranty Period for another four (4) years. Copyright 2003 Page 5 PIPTS101-10
44 Endosafe - Portable Test System User s Guide Charles River Laboratories will arrange transfer for the PTS instrument to the Company s technical services laboratory, and provide the client a loaner instrument during the period the instrument is being serviced (See Returned Goods Authorization Procedure below). The client shall be responsible for all freight charges associated with the returned instrument and the loaner instrument. ON DEMAND REPAIRS After the initial Warranty Period has ended, the client may request Charles River Laboratories to service or repair the PTS instrument without having the benefit of one of the above Service Agreements. Charles River Laboratories will arrange transfer of the PTS instrument to the Company s technical services laboratory, and provide the client a loaner instrument during the period On Demand repairs are being made (See Returned Goods Authorization Procedure below). The client shall be responsible for all freight charges associated with the returned instrument and the loaner instrument. RETURNED GOODS AUTHORIZATION PROCEDURE The client must obtain a Returned Goods Authorization Number from Charles River Laboratories before any items can be shipped to the Technical Services Department. All goods returned for repair, preventative maintenance, or calibration, whether under warranty or not, must be properly packed to avoid damage and clearly marked with the RGA number. WARRANTY REGISTRATION A Warranty Registration Card is provided in this User s Guide, and is also available online at Submitting the completed card will permit the Technical Services Department to more efficiently meet each client s needs. All warranty and service options are subject to the Terms and Conditions and procedures specified in the Service Agreement. For answers to questions about any of these components for the Endosafe - PTS or for additional product information, please contact Charles River Laboratories Technical Services Department: United States: 1023 Wappoo Road, Suite 43-B Charleston, SC USA (800) Europe: Copyright 2003 Page 6 PIPTS101-10
45 BATTERY INFORMATION Endosafe - Portable Test System User s Guide Operating the Reader The PTS Reader can operate on its own battery power or with the supplied AC power adapter. The AC power adapter also serves as the battery charger for the PTS Reader. The batteries enable the user to operate the PTS Reader onsite without the use of a power cord. The rechargeable lithium-ion batteries are installed at the factory and should be replaced only by the factory. The following procedures are presented so the user can experience the maximum useful performance and life from the batteries. The batteries should be fully charged (about 4 to 4.5 hours) before use. This will give approximately 6 to 8 hours of continuous usage. To charge the batteries, use only the AC power adapter supplied with the instrument. NOTE: Insert the power cord into any grounded 3-prong outlet, and then insert the DC plug into the rear of the PTS Reader. The AC power cord adapter displays a green LED when the cord is securely plugged into a wall outlet. If this light does not appear, try a different socket or make sure the power cord is properly inserted into the outlet. Never use a charger that is damaged. A desirable operating condition for the batteries is to be fully charged and then used to operate the PTS Reader on battery power until a low level of charge is reached best if fully discharged. Then repeat the full charge-full discharge usage cycle. Leaving the PTS on continuously with the external charger connected is not recommended. NOTE: If the PTS Reader is continuously connected to an external charger, the batteries will self-discharge approximately 10 to 15%, and then the charging circuit will activate and restore the charge. This will result in a shortened life span of the batteries. When operating on batteries, eventually a LOW BATTERY message will be displayed indicating that only one (1) or two (2) more tests can be performed before the battery charge is too low to initiate and complete another test. The AC power adapter must then be connected to continue operation. Testing can be resumed as the batteries are in the process of being charged. As the batteries approach their end of life (approximately 300 charge-discharge cycles), the number of tests that can be run on one (1) charge will diminish. The Charge Battery message will start to appear more often. The user will be alerted with the display and an audible beep every 5 minutes that there is a Battery Issue See User Manual. The alerts will also occur on system start-up but will not occur during a test. If the alerts are not heeded, and the batteries eventually reach full discharge, the PTS Reader will shut down, but will resume operation within 1 minute once the AC power adapter has been connected. Although operational with end-of-life batteries, the PTS Reader will be vulnerable to power interruptions. In all instances, when the batteries approach end of life, the PTS Reader should be returned to the factory for battery replacement. Copyright 2003 Page 7 PIPTS101-10
46 Endosafe - Portable Test System User s Guide If the batteries are to remain unused for extended periods of time, it is recommended that they be stored at a charge level of 30 to 70%; maximizing the overall life of the battery. KEYPAD OPERATION It is important to develop a thorough knowledge of the use of the PTS Reader keypad. Learning proper sequences for data entry will ensure your test procedures are successful, and that all necessary results are displayed at the conclusion of each test. The following overview of the PTS keypad operation covers all necessary functions and procedures for proper use of the PTS Reader during test operations. A. ON/OFF ON When using battery power, pressing the MENU key on the keypad will turn the instrument on. OFF Pressing the MENU key from the initial screen followed by the 5 key will turn the instrument off, conserving battery power. NOTE: Press CANCEL to return to the initial screen. B. MENU KEY 1. Pressing the Menu key initiates the Main Menu display on the PTS Reader s information screen. 2. Once Main Menu is displayed on the screen, Use the Menu key repeatedly to scroll through the separate, numbered headers that are available on the Main Menu: #1 Enter Test Inf The PTS Reader will prompt the user for all pertinent test information automatically when a cartridge is inserted. It is not necessary to Enter Test Info under this header. #2 - User Setup The majority of the options under this header are for factory use only. If selected, Action Denied will be displayed on the screen with an audible notification. The user has the capability of erasing all stored lot numbers under this heading. #3 - System Menu This header allows you to enter a variety of constants, such as: Time Menu, Date Menu, Languages, System Options, and setting the COM1 Baud rate. NOTE: Selecting any sub-header that is for factory use only will display Action Denied on the screen with an audible notification. #4 - Print Menu This header allows you to set options to govern the data printing process for your test results, and includes settings for: Printing the Last Test, Print all tests by a given date, Printing All Tests stored on the PTS Reader (up to 100 max.), Print 40 Character Line, A Display of # of Tests stored on the reader, Performing a Printer Test, and Erasing All Records. Copyright 2003 Page 8 PIPTS101-10
47 Endosafe - Portable Test System User s Guide NOTE: Once Erase All Records has been selected and confirmed, all records will be erased and irretrievable. If necessary, download desired data to PC, Seiko DPU-414 Printer, or Epson TM-U220D Printer to store prior to erasing all records (see pages 12-14). #5 System OFF Selecting this Main Menu option will turn the PTS reader off. #6 System Reset Selecting this Main Menu option will automatically reboot the test system. #7 System Info This header of the Main Menu allows you to view system information, such as the serial number, and the system software version. Becoming familiar with available options from each of the numbered headers of the Main Menu, allows you to proceed with greater speed and confidence as you perform your tests and specific data collection. Copyright 2003 Page 9 PIPTS101-10
48 Endosafe - Portable Test System User s Guide C. EXAMPLE OF KEYPAD OPERATION Setting the Time on the PTS Reader: 1. Turn the instrument on by pressing the MENU key The PTS Reader displays Insert Cartridge after Systems Self Test 2. Press the MENU key The PTS Reader displays Main Menu and 1-Enter Test Inf 3. Press MENU two (2) additional times for 3-System Menu, and then press 3 The PTS Reader displays System Menu and 1- Display Date Time 4. Press 3 to select the Time Menu 5. Use the MENU key to scroll through the options governing the time setting feature: 1 Set Time, 2 12 Hour, 3 24 Hour 6. For each of the 3 Time Menu items, you can press the ENTER key to enter the appropriate information regarding the time settings and preferences you want the system to use 7. For example, if you press ENTER when the PTS Reader displays 1 Set Time, you will see the default time on the PTS Reader screen. A cursor will appear below the first number, which allows you to enter a new number in that position to format the correct time. Simply move the cursor left or right using the arrow keys, and press the numeral keys on the keypad to enter the correct time 8. Once you have entered the appropriate information, press the ENTER key The PTS Reader displays your entry and Stored, to indicate the data was stored successfully Copyright 2003 Page 10 PIPTS101-10
49 Endosafe - Portable Test System User s Guide D. MENU SHORTCUTS FOR PTS READER KEYPAD The following Menu Shortcuts diagrams the Menu options and their headings and subheadings. Using the table, you can go directly to any of the available menu fields by pressing the MENU key and the appropriate menu item code number. M2 M24 M25 M3 M31 M331 M332 M333 M321 M322 M323 M41 M42 M43 M44 M45 M46 M47 M5 M6 M7 MAIN MENU USER SETUP 4-Erase Lot Numb 5-Channel EU YES/NO 3-System Menu SYSTEM MENU 1- Displ DateTime TIME MENU 1-Set Time 2-12 Hour 3-24 Hour DATE MENU 1-Set Date 2-MON/DAY/YEAR 3-DAY/MON/YEAR PRINT MENU 1-Print Last Tst 2-Print All Tsts on given date 3-Print All Tsts 4-Print 40-Character format 5-#Tests Stored: 6-Printer Test 7-Erase All Recs 5-System OFF 6-System Reset 7-System Info E. CANCEL KEY Pressing the CANCEL key at any time during data entry to the PTS Reader will return the display to the previous numbered header of the Main Menu. If you continue to press the CANCEL key, the display will cycle over to the Main Menu display, and subsequently out of all menus, at which point the PTS Reader will display Insert Cartridge. At this point, if all necessary information has been entered to the PTS Reader for the test setup, testing can begin. Copyright 2003 Page 11 PIPTS101-10
50 Endosafe - Portable Test System User s Guide Reader Output Downloading Output to PC using the PTS Logger Software 1. Insert the PTS Logger software CD into your PC and download to PC. 2. Connect Cable Serial port connection to PC and RJ45 plug into back of PTS Reader (cable is provided with PTS System). 3. Select Connect then choose the com port your PTS Reader is connected to in the Setup communications window. Click on the Connect button to connect to PTS Reader. 4. Select Log then Open/Close Log to open the log. 5. Press the MENU key on the PTS Reader keypad. Select 4 for the Print Menu. The options for downloading data to the PC are: 1-Print Last Test 2-Print by Date 3-Print all Tests Selecting one (1) of the above three (3) choices to print will automatically download results to the PTS Logger software if connected as described above. Copyright 2003 Page 12 PIPTS101-10
51 Endosafe - Portable Test System User s Guide 6. Select Log then View to view data in notepad format. Note: The data will automatically be sent to an unnamed Notepad file within the PTS Logger folder. If this file remains in the PTS Logger folder, the next set of data will be added to the bottom of this file. It is recommended that you either rename the Notepad file and move it to another folder or copy the data into a Word file and delete the notepad file. Printing with the DPU-414 Seiko Thermal Printer or the Epson TM- U220D Printer 1. Setup printer according to owner s manual instructions. 2. Connect Cable Serial port connection to Printer and RJ45 plug in back of the PTS Reader (Cable is provided with PTS System). 3. Press the MENU key on the PTS Reader keypad. 4. Select 4 for the Print Menu. The options for downloading data to the Printer are: 1-Print Last Test 2-Print by Date 3-Print all Tests 5. Selecting one (1) of the above three (3) choices on the PTS Reader keypad will send results to the connected Printer. 6. If the user desires to also download data to a PC, follow instructions above for Downloading Output to PC using the PTS Logger software. Copyright 2003 Page 13 PIPTS101-10
52 Endosafe - Portable Test System User s Guide Using EndoScan-V with Endosafe - PTS for data acquisition. 1. Insert the EndoScan-V CD into the CD drive. 2. Install EndoScan-V software. 3. Upon successful installation and registration of the software select PTS/MCS as the reader type from the reader options dialog box. 4. Once successfully connected to a PTS Reader the following screen will appear. While connected using EndoScan-V keyboard operations of the PTS are disabled. Copyright 2003 Page 14 PIPTS101-10
53 Endosafe - Portable Test System User s Guide 5. Enter the Cartridge lot number. The cartridge lot number will be stored for later use. 6. Enter the Calibration code. The Calibration code will be stored for later use. 7. Enter product information by either typing directly into the box or by dragging information from the product database into the dialog box. 8. Enter the dilution in the form of 1:x ml/ml or x units/ml for concentration. 9. Insert the cartridge. 10. Select the Send Setup button to send information to the PTS Reader. 11. Once the Add sample and Click start message is displayed dispense 25 μl into each well of the test cartridge and click start on the screen. 12. While in progress the screen will display messages showing the current progress. Copyright 2003 Page 15 PIPTS101-10
54 Endosafe - Portable Test System User s Guide 13. Upon completion of the assay, a message will be displayed instructing the user to remove the cartridge and a screen similar to the one below containing data for the assay will be displayed. 14. Select the report option to print a report for this assay. 15. Select the save or save as option to save the assay as an EndoScan-V file if Autoname is not selected. 16. To return the PTS to keypad simply disconnect the serial cable from the rear of the PTS unit. The unit will perform a system self-check and return to keypad operations. Copyright 2003 Page 16 PIPTS101-10
55 Endosafe - Portable Test System User s Guide Troubleshooting Listed in the table below are various Error or Fault Messages and a description of the corresponding cause of the error or fault. One or more LONG audible tones will precede each error message. Any fault listed here will prevent the PTS Reader from running a test. Unless it is a hardware problem, the only way to clear an error message is to turn the PTS Reader OFF and back ON again. MESSAGE A2D SETUP TEMP TOO LOW TEMP TOO HI MOTOR NOT HOME CLOCK FAULT Computing Error Charge Battery Battery Fault UNKNOWN One or more test LEDs out of spec Abort Test CANCEL OR ENTER Sample Not Seen flashes during test DESCRIPTION Photo A/D set up did not complete Cartridge temperature is too low Cartridge temperature is too high Cannot run pump motor Cannot read Real Time Clock Divide by 0 or range error Batteries too weak to run tests Battery charging problem Any unspecified system fault One or more of the LEDs does not have sufficient intensity to run the test properly Sample was not detected in one or more of the optical cells If any of these or other problems persists, contact Charles River Laboratories Technical Service: United States: 1023 Wappoo Road, Suite 43-B Charleston, SC USA (800) Europe: Fax: (843) [email protected] Copyright 2003 Page 17 PIPTS101-10
56 Endosafe - Portable Test System User s Guide Appendix A Endosafe - PTS Endotoxin PERFORMING ROUTINE ENDOTOXIN TEST A routine PTS LAL assay is conducted by following the simple prompts on the PTS instrument. The following represents a typical assay procedure: 1. Instrument Operation Press the MENU key on the PTS keypad to turn instrument on (Menu 5 turns instrument off). The PTS Reader initiates a SYSTEM SELF TEST as it heats up to 37 C this takes approximately 5 minutes. The PTS Reader displays SELF TEST OK and then INSERT CARTRIDGE. 2. Insert the Cartridge Note: Allow the cartridge to come to room temperature in pouch before use. Remove cartridge from pouch and insert with sample reservoirs facing up into slot at front of the PTS Reader. Press cartridge firmly into slot. 3. Enter Required Information Once the cartridge has been firmly inserted into the PTS Reader, the PTS Reader prompts the user to enter the following information: Enter OID (Operator ID) Enter Lot # (Cartridge Lot #) Enter Calibration Code (If the Calibration Code for the particular lot # has already been entered, the PTS Reader does not prompt for the code again. (To Erase all stored lot # s and corresponding calibration codes, select menu, 2, then 4 from the initial menu) Lot # (Confirms cartridge lot number entered) Enter Sample Lot # Enter Sample ID (Selecting and scrolling with the menu key under the sample ID Header allows for fifty (50) samples to be entered and stored) Enter Dilution Factor Note: All LAL Reagent Water (LRW) used to make product dilutions should be tested for the absence of detectable endotoxin. While the above information is being entered into the PTS Reader, the cartridge is being pre-warmed. Copyright 2003 PIPTS Page A1
57 Endosafe - Portable Test System User s Guide 4. Dispense the Sample Once all test information is entered, the PTS Reader displays: ADD SAMPLE; PRESS ENTER Pipette 25 μl of sample into all four (4) sample reservoirs of the inserted cartridge and press Enter on the PTS Reader keypad. Pumps draw sample aliquots into the test channels, thereby initiating the test. Results will be obtained in approximately 15 minutes. ENDOTOXIN TEST RESULTS 1. When the test is complete, the PTS Reader gives an audible notification that the assay is finished. 2. At the conclusion of the test, the endotoxin measurement and the assay acceptance criteria are displayed on the screen. 3. The PTS Reader display alternates between the following results: Sample EU/mL Sample %CV Spike EU/mL Spike %CV % Spike Recovery Remove Cartridge The PTS Reader continues to display the assay results until cartridge is removed. Copyright 2003 PIPTS Page A2
58 Endosafe - Portable Test System User s Guide RETRIEVING RESULTS OPTIONS 1. You can download results directly to your PC and retrieve from the designated location file. 2. You can use the Seiko DPU-414 Printer or the Epson TM-U220D Printer (available from Charles River Laboratories) to print the last test result, all results from a particular date, or up to a maximum of one hundred (100) stored test results. ********* ENDOSAFE Test Record ********* V7.12B 12/9/2008 DateTime:... 11:04:12AM Device: OperatorID:... KMULIK Cartridge:... Endotoxin Temperature:.. Start: 37.0C End: 37.0C Method:... KX-122 Cartridge Lot#: Cartridge Cal Code: Range: Range Time:... Sec: Onset Times:... > > Slope: Intercept: Dilution:... 1 Sample Lot: Sample ID:... LRW Sample Rxn Time CV: % Pass Spike Value: EU/mL Spike Rxn Time CV: % Pass Spike Recovery:... 92% Pass Test Suitability:... Pass Sample Value:... <0.050 EU/mL : Copyright 2003 PIPTS Page A3
59 Endosafe - Portable Test System User s Guide Appendix B Endosafe - PTS Rapid Micro Methods Gram ID PERFORMING ROUTINE GRAM ID TEST A routine Gram ID assay is conducted by preparing a bacterial suspension and following the simple prompts on the PTS Instrument. The following represents a typical assay procedure: The PTS Gram ID offers two (2) assay report formats. One assay result format will report the presence of either Gram-negative microorganisms, Yeast/Mold or Gram-positive microorganisms in approximately 7 minutes. The second assay result format will report the presence of Gram-negative and Gram-positive in approximately 3 minutes (this format does not differentiate between Gram-positive and Yeast/Mold isolates). Reference the Certificate of Analysis for the calibration code to be used for each assay result format. Sample Preparation: From an isolated culture, carefully remove colonies from agar surface and suspend cells in approximately 2 mls of LPS-free Saline or LRW and adjust to a 0.5 McFarland Equivalence Turbidity Standard. Note: Do not use cotton tip swabs of any kind during sample preparation. 1. Instrument Operation Press the MENU key on the PTS keypad to turn instrument on (Menu 5 turns instrument off). The PTS Reader initiates a SYSTEM SELF TEST as it heats up to 37 C this takes approximately 5 minutes. The PTS Reader displays SELF TEST OK and the INSERT CARTRIDGE. 2. Insert the Cartridge Remove cartridge from pouch and insert with sample reservoirs facing into slot at front of the PTS Reader. Press cartridge firmly into slot. Copyright 2003 PIPTS Page B1
60 Endosafe - Portable Test System User s Guide Note: If refrigerated, allow the cartridge to come to room temperature in pouch before use. 3. Enter Required Information Once the cartridge has been firmly inserted into the PTS Reader, the PTS Reader prompts the user to enter the following information: Enter OID (Operator ID) Enter Lot # (Cartridge Lot #) Note: If alternating between assay result formats, the user must delete all stored lot numbers in the PTS Reader before initiating a test. This is done by selecting Main Menu > #2 User Setup> #4 Erase Lot Numbers. Select Enter to erase all stored lot numbers Enter Calibration Code (If the Calibration Code for the particular lot # has already been entered, the PTS Reader does not prompt for the code again. After deleting stored lot numbers in the PTS Reader, the user will be prompted to enter in the calibration code) Lot # (Confirms cartridge lot number entered) Enter Sample #1 Lot # (This prompt is available for all four (4) samples) Enter Sample #1 ID (Selecting and scrolling with the menu key under the sample ID Header allows for fifty (50) samples to be entered and stored) While the above information is being entered into the PTS Reader, the cartridge is being pre-warmed. 4. Dispense the Sample Once all test information is entered, the PTS Reader displays: ADD SAMPLE; PRESS ENTER Pipette 25μL of first sample into one (1) sample reservoir of the inserted cartridge. Change pipette tip to avoid cross-contamination between each sample. After all four (4) sample reservoirs are filled, press Enter on the PTS Reader keypad. Pumps draw sample aliquots into the test channels, thereby initiating the test. Results will be obtained in approximately 7 minutes for Gram-negative, Gram-positive and Yeast/Mold identification and 3 minutes for Gramnegative and Gram-positive identification. Copyright 2003 PIPTS Page B2
61 Endosafe - Portable Test System User s Guide GRAM ID TEST RESULTS 1. When the test is complete, the PTS Reader gives an audible notification that the assay is finished. 2. At the conclusion of the test, the gram identification is displayed on the screen for each sample as Gram Negative, Gram Positive, Yeast / Mold. 3. The PTS Reader display alternates between the following results: Sample #1 Sample #2 Sample #3 Sample #4 Remove Cartridge The PTS Reader continues to display the assay results until the cartridge is removed. Copyright 2003 PIPTS Page B3
62 Endosafe - Portable Test System User s Guide RETRIEVING RESULTS OPTIONS 1. You can download results directly to your PC and retrieve from the designated location file. 2. You can use the Seiko DPU-414 Printer or the Epson TM-U220D Printer (available from Charles River Laboratories) to print the last test result, all results from a particular date, or up to a maximum of one hundred (100) stored test results. Example of Gram-negative or Gram-positive assay result format report: ********* ENDOSAFE Test Record ********* V7.12A 7/30/2008 DateTime:... 12:02:26PM Device: OperatorID:... DN Cartridge:... Gram ID Temperature:.. Start: 37.0C End: 37.0C Method:... GSX-401 Cartridge Lot#: Cartridge Cal Code: Onset Times:... <150 <150 >151 >151 Sample #1 Lot:...EC0012 Sample #1 ID:... TB-1 Sample #2 Lot:...EC0013 Sample #2 ID:... TB-2 Sample #3 Lot:...BC0004 Sample #3 ID:... FL-1 Sample #4 Lot:...BC0005 Sample #4 ID:... GL-1 Sample #1:... Gram Negative Sample #2:... Gram Negative Sample #3:... Gram Positive Sample #4:... Gram Positive Example of Gram-negative, Yeast/Mold or Gram-positive assay result format report: ********* ENDOSAFE Test Record ********* V7.12A 7/30/2008 DateTime:... 11:45:32AM Device: OperatorID:... DN Cartridge:... Gram ID Temperature:.. Start: 37.0C End: 37.0C Method:... GSX-401 Cartridge Lot#: Cartridge Cal Code: Onset Times: Sample #1 Lot:... EC0001 Sample #1 ID:... FL-2 Sample #2 Lot:... CA0002 Sample #2 ID:... FL-2 Sample #3 Lot:... CA0003 Sample #3 ID:... TB-3 Sample #4 Lot:... BC0006 Sample #4 ID:... GL-2 Sample #1:... Gram Negative Sample #2:... Yeast / Mold Sample #3:... Yeast / Mold Sample #4:... Gram Positive Copyright 2003 PIPTS Page B4
63 Endosafe - Portable Test System User s Guide Appendix C Endosafe - PTS Inhibition/Enhancement Screen PERFORMING INHIBITION/ENHANCEMENT TEST An Inhibition/Enhancement PTS LAL assay is conducted by following the simple prompts on the PTS instrument. Different dilutions of one (1) sample can be placed in each sample well to determine the non-interfering dilution or different samples can be placed in each well of one (1) test cartridge. The following represents a typical assay procedure: 1. Instrument Operation Press the MENU key on the PTS keypad to turn instrument on (Menu 5 turns instrument off). The PTS Reader initiates a SYSTEM SELF TEST as it heats up to 37 C this takes approximately 5 minutes. The PTS Reader displays SELF TEST OK and then INSERT CARTRIDGE. 2. Insert the Cartridge Remove cartridge from pouch and insert with sample reservoirs facing up into slot at front of the PTS Reader. Press cartridge firmly into slot. Note: Allow the cartridge to come to room temperature in pouch before use. 3. Enter Required Information Once the cartridge has been firmly inserted into the PTS Reader, the PTS Reader prompts the user to enter the following information: Enter OID (Operator ID) Enter Lot # (Cartridge Lot #) Enter Calibration Code (If the Calibration Code for the particular lot # has already been entered, the reader does not prompt for the code again. (To Erase all stored lot # s and corresponding calibration codes, select menu, 2, then 4 from the initial menu) Lot # (Confirms cartridge lot number entered) Enter Sample 1 Lot # (Sample 1 is designated as the well on the far left after cartridge is inserted into the PTS Reader) Copyright 2003 PIPTS Page C1
64 Endosafe - Portable Test System User s Guide Enter Sample 2 Lot # (Sample 2 is the next well to the right of sample 1) Enter Sample 3 Lot # (Sample 3 is the next well to the right of sample 2) Enter Sample 4 Lot # (Sample 4 is the last well on the right) Enter Sample 1 ID (Selecting and scrolling with the menu key under the sample ID Header allows for fifty (50) samples to be entered and stored) Enter Sample 2 ID Enter Sample 3 ID Enter Sample 4 ID Enter Dilution #1 Factor Enter Dilution #2 Factor Enter Dilution #3 Factor Enter Dilution #4 Factor Note: All LAL Reagent Water (LRW) used to make product dilutions should be tested for the absence of detectable endotoxin. While the above information is being entered into the PTS Reader, the cartridge is being pre-warmed. 4. Dispense the Sample Once all test information is entered, the PTS Reader displays: ADD SAMPLE; PRESS ENTER Pipette 25 μl of sample into four (4) sample reservoirs of the inserted cartridge and press Enter on the PTS Reader keypad. Pumps draw sample aliquots into the test channels, thereby initiating the test. Results will be obtained in approximately 15 minutes. Copyright 2003 PIPTS Page C2
65 Endosafe - Portable Test System User s Guide INHIBITION/ENHANCEMENT TEST RESULTS 1. When the test is complete, the PTS Reader gives an audible notification that the assay is finished. 2. At the conclusion of the test, the assay acceptance criteria are displayed on the screen. 3. The PTS Reader display alternates between the following results: Spike #1 Recovery Spike #2 Recovery Spike #3 Recovery Spike #4 Recovery The PTS Reader continues to display the assay results until cartridge is removed. Once the assay is complete and the results are noted, remove the cartridge promptly from the PTS Reader. Copyright 2003 PIPTS Page C3
66 Endosafe - Portable Test System User s Guide RETRIEVING RESULTS OPTIONS 1. You can download results directly to your PC and retrieve from the designated location file. 2. You can use the Seiko DPU-414 or the Epson TM-U220D Printer (available from Charles River Laboratories) to print the last test result, all results from a particular date, or up to a maximum of one hundred (100) stored test results. ********* ENDOSAFE Test Record ********* V7.12B 12/9/2008 DateTime:... 09:28:35AM Device: OperatorID:... KMULIK Cartridge:... I/E Screen Temperature:.. Start: 37.0C End: 37.0C Method:... KX-410 Cartridge Lot#: Cartridge Cal Code: Range: Range Time:... Sec: Onset Times:... > Slope: Intercept: Dilution #1: Sample #1 Lot: Sample #1 ID:... ETHANOL Dilution #2: Sample #2 Lot: Sample #2 ID:... ETHANOL Dilution #3: Sample #3 Lot: Sample #3 ID:... ETHANOL Dilution #4: Sample #4 Lot: Sample #4 ID:... ETHANOL Spike #1 Recovery:... < 11% Spike #2 Recovery:... 18% Spike #3 Recovery:... 72% Spike #4 Recovery:... 86% : Copyright 2003 PIPTS Page C4
67 Endosafe - Portable Test System User s Guide Appendix D Endosafe - PTS Rapid Micro Methods Glucan PERFORMING ROUTINE GLUCAN TEST The PTS Glucan assay is used for the detection of (1,3)- -D-Glucans. The Endosafe -PTS Glucan assay is intended for research purposes only and is not to be used for diagnostic testing purposes. A routine PTS Glucan assay is conducted by following the simple prompts on the PTS instrument. The following represents a typical assay procedure: 1. Instrument Operation Press the MENU key on the PTS keypad to turn instrument on (Menu 5 turns instrument off) The reader initiates a SYSTEM SELF TEST as it heats up to 37 C this takes approximately 5 minutes The reader displays SELF TEST OK and then INSERT CARTRIDGE 2. Insert the Cartridge Remove cartridge from pouch and insert with sample reservoirs facing up into slot at front of the PTS Reader. Press cartridge firmly into slot. Note: Allow the cartridge to come to room temperature in pouch before use. 3. Enter Required Information Once the cartridge has been firmly inserted into the reader, the reader prompts the user to enter the following information: Enter OID (Operator ID) Enter Lot # (Cartridge Lot #) Enter Calibration Code (If the Calibration Code for the particular lot # has already been entered, the reader does not prompt for the code again. (To Erase all stored lot # s and corresponding calibration codes, select menu, 2, then 4 from the initial menu) Lot # (Confirms cartridge lot number entered) Enter Sample Lot # Copyright 2003 PIPTS Page D1
68 Endosafe - Portable Test System User s Guide Enter Sample ID (Selecting and scrolling with the menu key under the sample ID Header allows for fifty (50) samples to be entered and stored) Enter Dilution Factor Note: All LAL Reagent Water (LRW) used to make product dilutions should be tested for the absence of detectable endotoxin and glucan. While the above information is being entered into the reader, the cartridge is being pre-warmed. 1. Dispense the sample Once all test information is entered, the reader displays: ADD SAMPLE; PRESS ENTER Pipette 25 μl of sample into four (4) sample reservoirs of the inserted cartridge and press Enter on the reader keypad Pumps draw sample aliquots into the test channels, thereby initiating the test Results will be obtained in approximately 30 minutes. GLUCAN TEST RESULTS 1. When the test is complete, the PTS Reader gives an audible notification that the assay is finished. 2. At the conclusion of the test, the glucan measurement and the assay acceptance criteria are displayed on the screen. 3. The reader display alternates between the following results: Sample pg/ml Value Sample %CV Spike pg/ml Value Spike %CV % Spike Recovery Remove Cartridge The reader continues to display the assay results until cartridge is removed. Once the assay is complete and the results are noted, remove the cartridge promptly from the reader. Copyright 2003 PIPTS Page D2
69 Endosafe - Portable Test System User s Guide RETRIEVING RESULTS OPTIONS 1. You can download results directly to your PC and retrieve from the designated location file. 2. You can use the Seiko DPU-414 or the Epson TM-U220D Printer (available from Charles River Laboratories) to print the last test result, all results from a particular date, or up to a maximum of one hundred (100) stored test results. Example of Glucan Test Report: ********* ENDOSAFE Test Record ********* V7.12B 12/9/2008 DateTime:... 08:59:40AM Device: OperatorID:... DN Cartridge:... PTS Glucan Temperature:.. Start: 37.0C End: 37.0C Method:... KX-122 Cartridge Lot#: Cartridge Cal Code: Range: Range Time:... Sec: Onset Times:... > > Slope: Intercept: Dilution:... 1 Sample Lot: Sample ID:... RD0901 Sample Rxn Time CV: % Pass Spike Value: pg/ml Spike Rxn Time CV: % Pass Spike Recovery:... 99% Pass Test Suitability:... Pass Sample Value:... <10.0 pg/ml : Copyright 2003 PIPTS Page D3
70 Endosafe - Portable Test System User s Guide Appendix E Abbreviations used in this User s Guide: PTS LAL FDA EU LRW Portable Test System Limulus Amebocyte Lysate Food and Drug Administration Endotoxin Unit LAL Reagent Water Copyright 2003 PIPTS Page E1
71 Endosafe - Portable Test System User s Guide Appendix F Decontamination Procedures Warning: Do not immerse the Endosafe PTS instrument, spray it with liquid, or use a wet cloth. Do not allow the cleaning solution to run into the interior of the Endosafe PTS instrument. If this happens, contact Charles River Laboratories Technical Service Department. Do not soak the keypad, this will cause damage. Important! Turn off and unplug the Endosafe PTS instrument for all decontamination and cleaning operations. 1. Turn off and unplug the Endosafe PTS instrument. 2. Moisten a cloth with a mild detergent solution. Do not soak the cloth. Wipe down all surfaces of the Endosafe PTS instrument. 3. Use a clean dry cloth to dry all wet surfaces. Copyright 2003 PIPTS Page F1
72 Instruções de Uso do Produto 2: Endosafe MCS (Multi-Cartridge System)
73 The Endosafe MCS Multi-Cartridge System USER S GUIDE Information Only 05-FEB Wappoo Road Suite 43-B, Charleston, SC Phone: Fax: [email protected]
74 Endosafe - Multi-Cartridge System User s Guide Table of Contents Safety Guidelines... 1 Endosafe - MCS System Overview Reader Specifications... 3 Cartridge Specifications... 4 Accessory Products... 4 Environmental Requirements Installation... 5 Controls and Signals Endosafe - MCS Service Programs Warranty with Purchase... 8 On Demand Repairs... 9 Returned Goods Authorization Procedure... 9 Support... 9 Using EndoScan-V with Endosafe -MCS for data acquisition...10 Troubleshooting Information Only 05-FEB-2014 Copyright 2003 PIMCS101-04
75 Endosafe - Multi-Cartridge System User s Guide Appendixes Appendix A Performing Routine Endotoxin Test... A1 Endotoxin Test Results... A2 Retrieving Results Options... A2 Appendix B Performing Routine Gram ID Test... B1 Gram ID Test Results... B3 Retrieving Results Options... B3 Appendix C Performing Inhibition/Enhancement Test... C1 Test Results... C3 Appendix D Performing Routine Glucan Test... D1 Glucan Test Results... D2 Retrieving Results Options... D2 Appendix E Abbreviations Used.... E1 Appendix F Decontamination Procedures.... F1 Information Only 05-FEB-2014 Copyright 2003 PIMCS101-04
76 Endosafe - Multi-Cartridge System User s Guide SAFETY FIRST WARNING CAUTION DO NOT use an AC power cord other than that which has been delivered with your system. Only plug the cord into a socket which is grounded. Using a different cord may cause fire leading to serious injury. DO NOT place MCS system around any liquids or use in a location where it could get wet or splashed. Liquids could cause fire or electric shock. IF the equipment is used in a manner not specified by the manufacturer, the protection provided by the equipment may be impaired. DO NOT open the instrument and tamper with the interior components. It does not contain any parts that can be maintained, repaired, or replaced by the user. Doing so may cause fire or electric shock. DO NOT allow liquids to spill into the cartridge chamber. Doing so may damage interior components and may cause fire or electric shock. DO NOT unplug AC power cord or PC cable by pulling cord/cable directly. Doing so may damage cord and cause injury. Handle the AC power cord and Serial Port Connector directly to unplug. Read this User s Guide carefully before using the Endosafe - MCS System. Understanding- the proper usage and operation is imperative for the safety of the User, as well as the protection of the Endosafe - MCS System. Information Only 05-FEB-2014 Copyright 2003 Page 1 PIMCS101-04
77 Endosafe - Multi-Cartridge System User s Guide Endosafe - MCS System Overview The Endosafe - MCS (Multi-Cartridge System) is a rapid test system which is comprised of a test cartridge along with a spectrophotometer. The Endosafe - MCS Endotoxin test utilizes existing FDA-licensed LAL formulations. The MCS provides quantitative LAL test results in approximately 15 minutes (see Appendix A for testing procedures and test results). All components required for testing samples with the MCS are listed below. Product Description Catalog # Quantity Endosafe - MCS kit MCS100 Each Reader and Integrated software Multi-Cartridge Multi- Integrated power supply Cartridge System Power cable User s Guide Endosafe - Licensed Cartridge 0.1 EU/mL sensitivity PTS201F 1/pack Pack 0.05 EU/mL sensitivity PTS2005F (10 cartridges per package) 0.01 EU/mL sensitivity PTS2001F EU/mL sensitivity PTS20005F Endosafe - Unlicensed Cartridge Pack (10 cartridges per package) Endosafe -Licensed Multi Pack Cartridge (25 cartridges per package) Endosafe -Licensed Multi Pack Cartridge (50 cartridges per package) Endosafe -Licensed Multi Pack Cartridge (100 cartridges per package) Endosafe - PTS Inhibition/Enhancement Screening Cartridge Pack (10 cartridges per package) 0.1 EU/mL sensitivity 0.05 EU/mL sensitivity 0.01 EU/mL sensitivity 5 to 0.05 EU/mL sensitivity 1 to 0.01 EU/mL sensitivity 0.5 to EU/mL sensitivity 5 to 0.05 EU/mL sensitivity 1 to 0.01 EU/mL sensitivity 5 to 0.05 EU/mL sensitivity 1 to 0.01 EU/mL sensitivity Inhibition/Enhancement Screening PTS201 PTS2005 PTS2001 PTS5505F PTS5501F PTS55005F PTS1105F PTS1101F PTS1105F PTS1101F PTS220 1/pack (5ct. 5pk.) (5ct. 10pk.) (10ct. 10pk.) 1/pack Information Only 05-FEB-2014 Endosafe - PTS RMM Gram ID Cartridge (10 cartridges per package) Endosafe - PTS RMM Glucan Cartridge (10 cartridges per package) Gram ID LRMMGI100 1/pack (1 3)-β-D-Glucan RMMGS1000 1/pack Copyright 2003 Page 2 PIMCS101-04
78 Endosafe - Multi-Cartridge System User s Guide Endosafe - MCS Reader Specifications Temperature control: 37º C + 1º C Data interface: USB port Printer support: Windows supported printer Data storage: Files are stored on PC Dimensions: 375mm (D) x 370mm (W) x 105mm (H) Power supply unit: VAC, 50/60Hz, 1.75 A max. Power supply on board: 12 V DC, 5A Fuse: 2x T2.5A/L 250V, 5x20mm Warm-up time: Approximately 5-10 minutes from 20º C start Environmental Conditions: o Operating Temperature 15 to 27 C o Humidity 5% to 80% noncondensing o Indoor use o Altitude : normal environmental conditions Weight: 8.3 kg Information Only 05-FEB-2014 Copyright 2003 Page 3 PIMCS101-04
79 Endosafe - Multi-Cartridge System User s Guide Endosafe - MCS Cartridge Specifications Reference package insert for storage requirements Endotoxin Sensitivity: EU/mL range, EU/mL range, EU/mL range, EU/mL range (see Appendix A) Rapid Micro Methods Gram ID: Archived parameters for Gram-negative, Grampositive and Yeast/Mold isolate identification (see Appendix B) Inhibition/Enhancement Screening (see Appendix C) Rapid Micro Methods Glucan (see Appendix D) Acceptable temperature: 37º C + 1º C Sample requirement: 25 µl sample per channel NOTE: Sample must be delivered to each sample port without contamination or cross contamination between samples. Endosafe - MCS Accessory Products Product Description Code Endosafe - MCS mini pipettor 25 µl disposable unit PTS400 Eppendorf * pipette tips *Eppendorf is a registered trademark of Brinkmann Multiple patents pending. 200 µl, individually wrapped, validated to EU/mL D200 Information Only 05-FEB-2014 Copyright 2003 Page 4 PIMCS101-04
80 Endosafe - Multi-Cartridge System User s Guide Endosafe - MCS Reader Environmental Requirements Surface: flat, level surface, vibration free Surroundings: free from dust, solvents and acidics vapours, and out of direct sunlight Operating Temperature: 15 to 27 C Humidity: 5% to 80% noncondensing Endosafe - MCS Reader Installation The MCS reader comes as a ready to use device, so no further installations on the hardware are necessary. Simply connect the MCS reader to the computer with the provided USB cable. The Type B connector goes to the reader and the Type A connector to the PC. Provide the MCS reader with power by connecting the provided power cable to the entry module of the reader and the grounded mains outlet. DO NOT USE UNGROUNDED POWER OUTLETS. During operation of the MCS reader the lateral venting slots must not be blocked. Also the cooling fan in the back of the MCS reader must not be blocked at any time. Please ensure a 50 mm safety clearance on the left, right and back side of the instrument. Endosafe - MCS Reader Controls and Signals Front/Side view: Lateral Venting Slots Cartridge Slot 1 Cartridge Slot 2 Information Only 05-FEB-2014 Cartridge Slot 3 Cartridge Slot 4 Cartridge Slot 5 Blue Power ON LED Copyright 2003 Page 5 PIMCS101-04
81 Endosafe - Multi-Cartridge System User s Guide Side view: Back left view: Fuse Holder ON/OFF Switch Back right view: Power Entry Module USB Power Adapter Plug Information Only 05-FEB-2014 Red USB Power LED Copyright 2003 Page 6 PIMCS USB Connector Type B
82 Endosafe - Multi-Cartridge System User s Guide After the warm-up time you will notice a short beep for each of the five (5) cartridge slots to indicate that the temperature has reached 37 degrees Celsius. The MCS reader is then ready to be operated through EndoScan-V. Cartridges can now be used according to our cartridge package inserts. Please hold the cartridges as shown below. Touch only the handle end of the cartridge (curved edges) to place the cartridge into the reader. Information Only 05-FEB-2014 Copyright 2003 Page 7 PIMCS101-04
83 Endosafe - Multi-Cartridge System User s Guide Insert cartridge firmly into the slot at the front of the reader with the sample reservoirs facing up. Endosafe - MCS Service Programs WARRANTY with PURCHASE Charles River Laboratories, Inc. warrants the Endosafe - MCS instrument will be delivered in a functional state and free from defect, and will meet stated specifications for a period of one (1) year. The warranty period will start on the date of shipment by Charles River Laboratories, Inc. During the warranty period, Charles River Laboratories, Inc. will repair the instrument at no charge to the client for parts and labor. If repairs are needed, Charles River Laboratories, Inc. will provide the client a loaner instrument during the period the instrument is being repaired. The client shall be responsible for all freight charges associated with the returned instrument and the loaner instrument. Opening the case or tampering with the interior components of the reader will invalidate warranty provisions. Additional service contracts are available as follows: Information Only 05-FEB-2014 Service MCS IQ,OQ,PQ Qualification On Demand Repairs Code MCS502 MCS503 All warranty and service options are subject to the Terms and Conditions and procedures specified in the Service Agreement. Copyright 2003 Page 8 PIMCS101-04
84 Endosafe - Multi-Cartridge System User s Guide ON DEMAND REPAIRS After the initial Warranty Period has ended, the client may request Charles River Laboratories, Inc. to service or repair the MCS. Charles River Laboratories, Inc. will arrange transfer of the MCS instrument to the Company s Technical Services Laboratory, and provide the client a loaner instrument during the period On Demand repairs are being made (See Returned Goods Authorization Procedure below). The client shall be responsible for all freight charges associated with the returned instrument and the loaner instrument. RETURNED GOODS AUTHORIZATION PROCEDURE The client must obtain a Returned Goods Authorization Number from Charles River Laboratories, Inc. before any items can be shipped to the Technical Services Department. All goods returned for repair, preventative maintenance, or calibration, whether under warranty or not, must be properly packed to avoid damage and clearly marked with the RGA number. SUPPORT For answers to questions about any of these components for the Endosafe - MCS or for additional product information, please contact Charles River Laboratories, Inc. Technical Services Department: United States: 1023 Wappoo Road, Suite 43-B Charleston, SC USA (800) Europe: Information Only 05-FEB-2014 Copyright 2003 Page 9 PIMCS101-04
85 Endosafe - Multi-Cartridge System User s Guide Operating the Reader Using EndoScan-V with Endosafe -MCS for data acquisition. 1. Insert the EndoScan-V CD into the CD drive. 2. Install EndoScan-V software. 3. Upon successful installation and registration of the software select PTS/MCS as the reader type from the reader options dialog box. 4. Once successfully connected to a MCS Reader the following screen will appear. This will show all available bays for each instrument connected. Information Only 05-FEB-2014 Copyright 2003 Page 10 PIMCS101-04
86 Endosafe - Multi-Cartridge System User s Guide 5. Select Tools/Options/Calculations and change CV Limit Products and CV Limits Standards to 25. Change Spike recovery range to 50 to Enter the Cartridge lot number. The Cartridge lot number will be stored for later use. 7. Enter the Calibration code. The Calibration code will be stored for later use. 8. Enter product information by either typing directly into the box or by dragging information from the product database into the dialog box. 9. Enter the concentration or dilution. Dilution should be in a format of #:#. 10.Insert the cartridge into the bay. 11.Select the Send Setup button to send information to the MCS Reader. Information Only 05-FEB-2014 Copyright 2003 Page 11 PIMCS101-04
87 Endosafe - Multi-Cartridge System User s Guide 12. Once the Add sample and Click start message is displayed dispense 25 µl into each well of the test cartridge. 13. Repeat steps 5 through 12 for each bay that will be used. 14. While in progress the screen will display messages showing the current status. Information Only 05-FEB-2014 Copyright 2003 Page 12 PIMCS101-04
88 Endosafe - Multi-Cartridge System User s Guide 15. Upon completion of the assay a message will be displayed instructing the user to remove the cartridge and a screen similar to the one below containing data for the assay will be displayed. Information Only 05-FEB Select the report option to print a report for this assay. 17. Select the save or save as option to save the assay as an EndoScan-V file. 18. The screen will display a message asking to save an individual assay or all assays that have been completed. 19. Select the desired method. 20. Assay is now saved as an EndoScan-V file. Copyright 2003 Page 13 PIMCS101-04
89 Endosafe - Multi-Cartridge System User s Guide Troubleshooting Listed in the table below are various Error or Fault Messages and a description of the corresponding cause of the error or fault. One or more LONG audible tones will precede each error message. Any fault listed here will prevent the reader from running a test. Unless it is a hardware problem, the only way to clear an error message is to turn the reader OFF and back ON again. MESSAGE A2D SETUP TEMP TOO LOW TEMP TOO HI MOTOR NOT HOME CLOCK FAULT Computing Error UNKNOWN One or more test LEDs out of spec Abort Test CANCEL OR ENTER Sample Not Seen flashes during test DESCRIPTION Photo A/D setup did not complete Cartridge temperature is too low Cartridge temperature is too high Cannot run pump motor Cannot read Real Time Clock Divide by 0 or range error Any unspecified system fault One or more of the LEDs does not have sufficient intensity to run the test properly Sample was not detected in one or more of the optical cells If any of these or other problems persists, contact Charles River Laboratories, Inc. Technical Service: United States: 1023 Wappoo Road, Suite 43-B Charelston, SC USA Phone : (800) or (843) Fax: (843) [email protected] Information Only 05-FEB-2014 Europe: Copyright 2003 Page 14 PIMCS101-04
90 Endosafe - Multi-Cartridge System User s Guide Appendix A Endosafe - MCS Endotoxin PERFORMING ROUTINE TEST A routine MCS LAL assay is conducted by following the setup procedure. The following represents a typical assay procedure: 1. Instrument Operation Open the EndoScan-V software Verify the screen contains a panel for each bay of the instrument 2. Insert the Cartridge Note: Allow the cartridge to come to room temperature in pouch before use. Remove cartridge from pouch and insert with sample reservoirs facing up into one of the slots at front of the MCS Reader Press cartridge firmly into slot 3. Enter Required Information Once the cartridge has been firmly inserted into the reader, the software prompts the user to enter the following information: Enter Cartridge Lot # Enter Calibration Code (If the Calibration Code for the particular lot # has already been entered, the software automatically inputs this information until the lot numbers and calibration codes are erased by the user). Enter Product (Enter a sample ID or drag and drop information from the database). Enter Product Lot# Enter Dilution Factor or Concentration. Dilution factor entered in format #:#. Select the Send Setup box on the screen to transfer the product information to the MCS Reader. Information Only 05-FEB-2014 Note: All LAL Reagent Water (LRW) used to make product dilutions should be tested for the absence of detectable endotoxin. While the above information is being entered into the reader, the cartridge is being pre-warmed. Copyright 2003 PIMCS Page A1
91 Endosafe - Multi-Cartridge System User s Guide 4. Dispense the Sample Once all test information is entered, the software displays: Add Sample and click Start Pipette 25 µl of sample into all four (4) sample reservoirs of the inserted cartridge and select the start assay button on the panel. Pumps draw sample aliquots into the test channels, thereby initiating the test Results will be obtained in approximately 15 minutes TEST RESULTS 1. When the test is complete, the results will be displayed on screen. The software continues to display the assay results until the new assay button is selected. RETRIEVING RESULTS OPTIONS 1. Select Report from the file menu or the printer icon from the toolbar. The assay results will be shown in a report format. 2. Results may be printed to any Windows compatible printer. Information Only 05-FEB-2014 Copyright 2003 PIMCS Page A2
92 Endosafe - Multi-Cartridge System User s Guide Appendix B Endosafe - MCS Rapid Micro Methods Gram ID PERFORMING ROUTINE GRAM ID TEST A routine Gram ID assay is conducted by preparing a bacterial suspension and following the simple prompts on the EndoScan-V software. The following represents a typical assay procedure: The MCS Gram ID offers two (2) assay report formats. One assay result format will report the presence of either Gram-negative microorganisms, Yeast/Mold or Gram-positive microorganisms in approximately 7 minutes. The second assay result format will report the presence of Gram-negative and Gram-positive in approximately 3 minutes (this format does not differentiate between Gram-positive and Yeast/Mold isolates). Reference the Certificate of Analysis for the calibration code to be used for each assay result format. Sample Preparation: From an isolated culture, carefully remove colonies from agar surface and suspend cells in approximately 2 mls of LPS-free Saline or LRW and adjust to a 0.5 McFarland Equivalence Turbidity Standard. Note: Do not use cotton tip swabs of any kind during sample preparation. 1. Instrument Operation Open the EndoScan-V software Verify the screen contains a panel for each bay of the instrument 2. Insert the Cartridge Information Only 05-FEB-2014 Note: Allow the cartridge to come to room temperature in pouch before use. Remove cartridge from pouch and insert with sample reservoirs facing up into one of the slots at front of the MCS Reader Press cartridge firmly into slot Copyright 2003 PIMCS Page B1
93 Endosafe - Multi-Cartridge System User s Guide 3. Enter Required Information Once the cartridge has been firmly inserted into the reader, the software prompts the user to enter the following information: Enter Cartridge Lot # Enter Calibration Code (If the Calibration Code for the particular lot # has already been entered, the software automatically inputs this information until the lot numbers and calibration codes are erased by the user). Enter Product (Enter a sample ID or drag and drop information from the database. Repeat procedure 3 times and separate sample IDs by a semi-colon). Up to 4 separate samples can be tested. Enter Product Lot# (Separate product Lots# by a semi-colon). Enter Dilution Factor Select the Send Setup box on the screen to transfer the product information to the MCS Reader. Note: All LAL Reagent Water (LRW) used to make product dilutions should be tested for the absence of detectable endotoxin. While the above information is being entered into the reader, the cartridge is being pre-warmed. 4. Dispense the Sample Once all test information is entered, the software displays: Add Sample and click Start Pipette 25 µl of sample into all four (4) sample reservoirs of the inserted cartridge and select the start assay button on the panel. Pumps draw sample aliquots into the test channels, thereby initiating the test Information Only 05-FEB-2014 Results will be obtained in approximately 7 minutes for Gramnegative, Gram-positive and Yeast/Mold identification and 3 minutes for Gram-negative and Gram-positive identification. Copyright 2003 PIMCS Page B2
94 Endosafe - Multi-Cartridge System User s Guide GRAM ID TEST RESULTS 1. When the test is complete, the results will be displayed on bay screen as +, - and Y/M. 2. At the conclusion of the test, the gram identification is displayed on the screen for each sample as Gram Negative, Gram Positive, Yeast / Mold. The software continues to display the assay results until the new assay button is selected. RETRIEVING RESULTS OPTIONS 1. Select Report from the file menu or the printer icon from the toolbar. The assay results will be shown in a report format. 2. Results may be printed to any Windows compatible printer. Information Only 05-FEB-2014 Copyright 2003 PIMCS Page B3
95 Endosafe - Multi-Cartridge System User s Guide Appendix C Endosafe - MCS Inhibition/Enhancement Screen PERFORMING INHIBITION/ENHANCEMENT TEST An Inhibition/Enhancement MCS LAL assay is conducted by following the simple setup procedures. Different dilutions of one (1) sample can be placed in each sample well to determine the non-interfering dilution or different samples can be placed in each well of one (1) test cartridge. The following represents a typical assay procedure: 1. Instrument Operation Open the EndoScan-V software Verify the screen contain a panel for each bay of the instrument 2. Insert the Cartridge Remove cartridge from pouch and insert with sample reservoirs facing up into one (1) of the bays at front of the MCS Reader Press cartridge firmly into slot Note: Allow the cartridge to come to room temperature in pouch before use. 3. Enter Required Information Once the cartridge has been firmly inserted into the reader, enter the following information into the software: Enter Lot # (Cartridge Lot #) Enter Calibration Code (If the Calibration Code for the particular lot # has already been entered, the software automatically inputs this information until the lot numbers and calibration codes are erased by the user.) Enter Product (If testing different products then enter the product information for each channel separated by a semi-colon. Sample 1 is designated as the well on the far left after cartridge is inserted into the reader. If testing the same product in all four (4) channels then it is only necessary to enter the product once.) Information Only 05-FEB-2014 Copyright 2003 PIMCS Page C1
96 Endosafe - Multi-Cartridge System User s Guide EnterProduct Lot # (If testing different lots of product then enter lot # separated by a semi colon. If testing the same lot # in all four (4) channels then it is only necessary to enter the lot # once.) Enter Sample 1 ID (Selecting and scrolling with the menu key under the sample ID Header allows for fifty (50) samples to be entered and stored.) Enter Conc/Dilution (If testing different concentrations or dilutions then enter each concentration/dilution separated by a semi colon. For example, enter 1:1; 1:10; 1:100; 1:1000 if testing these dilutions. Select the unit of measure using the arrow down button.) Note: All LAL Reagent Water (LRW) used to make product dilutions should be tested for the absence of detectable endotoxin. While the above information is being entered into the reader, the cartridge is being pre-warmed. 4. Dispense the sample Once all test information is entered, the software displays: ADD SAMPLE and click Start Pipette 25 µl of sample into four (4) sample reservoirs of the inserted cartridge and press the start button. (Be sure to change tips between each dilution or sample) Pumps draw sample aliquots into the test channels, thereby initiating the test Results will be obtained in approximately 15 minutes Information Only 05-FEB-2014 Copyright 2003 PIMCS Page C2
97 Endosafe - Multi-Cartridge System User s Guide TEST RESULTS 1. When the test is complete, the MCS Reader gives an audible notification that the assay is finished 2. At the conclusion of the test, the assay acceptance criteria are displayed on the screen 3. The software displays the following results: Spike #1 Recovery Spike #2 Recovery Spike #3 Recovery Spike #4 Recovery When the test is complete the results will be displayed on the screen. The software continues to display the assay results until the new assay button is selected. Information Only 05-FEB-2014 Copyright 2003 PIMCS Page C3
98 Endosafe - Multi-Cartridge System User s Guide Appendix D Endosafe - MCS Rapid Micro Methods Glucan PERFORMING ROUTINE GLUCAN TEST The PTS Glucan assay is used for the detection of (1,3)-β-D-Glucans. The Endosafe - MCS Glucan assay is intended for research purposes only and is not to be used for diagnostic testing purposes. A routine MCS Glucan assay is conducted by following the simple prompts on the EndoScan-V software. The following represents a typical assay procedure: 1. Instrument Operation Open the EndoScan-V software Verify the screen contains a panel for each bay of the instrument 2. Insert the Cartridge Remove cartridge from pouch and insert with sample reservoirs facing up into slot at front of the MCS Reader Press cartridge firmly into slot Note: Allow the cartridge to come to room temperature in pouch before use. 3. Enter Required Information Once the cartridge has been firmly inserted into the reader, the software prompts the user to enter the following information: Enter Lot # (Cartridge Lot #) Enter Calibration Code (If the Calibration Code for the particular lot # has already been entered, the software automatically inputs this information until the lot numbers and calibration codes are erased by the user). Enter Product (Enter a sample ID or drag and drop information from the database). Enter Product Lot# Enter Dilution Factor or Concentration Select the Send Setup Box on the screen to transfer the product information to the MCS Reader. Information Only 05-FEB-2014 Copyright 2003 PIMCS Page D1
99 Endosafe - Multi-Cartridge System User s Guide Note: All LAL Reagent Water (LRW) used to make product dilutions should be tested for the absence of detectable endotoxin and glucan. While the above information is being entered into the reader, the cartridge is being pre-warmed. 2. Dispense the sample Once all test information is entered, the reader displays: ADD SAMPLE and click Start Pipette 25 µl of sample into four (4) sample reservoirs of the inserted cartridge and select the start assay button on the panel. Pumps draw sample aliquots into the test channels, thereby initiating the test Results will be obtained in approximately 30 minutes. GLUCAN TEST RESULTS 1. When the test is complete, the MCS Reader gives an audible notification that the assay is finished and the results will be displayed on the screen. The software continues to display the assay results until the new assay button is selected. RETRIEVING RESULTS OPTIONS 1. Select Report from the file menu or the printer icon from the toolbar. The assay results will be shown in a report format. 2. Results may be printed to any Windows compatible printer. Information Only 05-FEB-2014 Copyright 2003 PIMCS Page D2
100 Endosafe - Multi-Cartridge System User s Guide Appendix E Abbreviations used in this User s Guide: MCS LAL FDA EU LRW Bay Multi-Cartridge Multi-Cartridge System Limulus Amebocyte Lysate Food and Drug Administration Endotoxin Unit LAL Reagent Water Slot on the face of the MCS corresponding to an individual testing device Information Only 05-FEB-2014 Copyright 2003 PIMCS Page E1
101 Appendix F Decontamination Procedures Endosafe - Multi-Cartridge System User s Guide Warning: Do not immerse the instrument, spray it with liquid or use a wet cloth. Do not allow the cleaning solution to run into the interior of the instrument. If this happens contact Charles River Laboratories, Inc. Technical Service Department. Important! Turn off and unplug the instrument for all decontamination and cleaning operations. 1. Turn off an unplug instrument. 2. Moisten a cloth with a mild detergent solution. Do not soak the cloth. Wipe down all surfaces of the instrument. 3. Use a clean dry cloth to dry all wet surfaces. Information Only 05-FEB-2014 Copyright 2003 PIMCS Page F1
102 Instruções de Uso do Produto 3: Endosafe Nexus
103 2014 Charles River Laboratories International, Inc. Endosafe Nexus Manual
104 Table of Contents 1 Before Starting Starting the Software & Login Dialog Box Main Window Endosafe Nexus System Controls New Assay Icon Multi-Cartridge System Connection (Endosafe -MCS ) Nimbus Robot Connection Assay Configuration Shift and Scan Barcode Review Barcode Product Area Dilutions Automation Process Assay Real Time Status Assay Run Status Pause Functionality Nimbus Movements Preparing the dilution as a background task Assay Real Time Status of the Bays Starting a new automation cycle Configuration Plate File Path Summary Report File Path Export File Path Configuration Template Export Files Administration Maintain Cartridge Data Maintain Reagent Locations Report Assay Report Preview Button Report Navigation Report Search... 32
105 3.4.5 Report Zoom Export File Button Print Button Button Command Usage Log Report Nimbus Report View Log Nimbus Log Command Usage Log Help Exit... 37
106 1 Before Starting Deck configuration and basic considerations before starting: 1. Turn on Endosafe-MCS, Nimbus robot and laptop 2. Confirm waste container on deck is empty and is securely in place 3. Add 300uL and 1000uL tips to Nexus deck 4. Open multipack cartridge pack and add to cartridge clip starting from the far left channel and moving left to right. Be sure to fill up cartridge channel completely (20 cartridges) before moving to next channel. Cartridges must be used within validated time period. 5. Take two desiccants from multi-pack containers and add to space provided in cartridge clip (may need to fold desiccants to fit). 6. Place lid on cartridge clip and place clip onto Nexus deck. Ensure clip and dispenser are securely in place. 7. Load samples into sample racks and insert racks onto deck with the open slots facing the scanner. 8. If utilizing the Nexus automated dilution feature, make sure the Reagents are loaded into the Reagent Rack, and that there are unused Dilution tubes in the Dilution Tube Rack. 9. Ensure there are no obstructions within robotic field of motion and that no items are placed on top of the MCS. 10. Open Nexus software and follow instructions below to begin running assays.
107 2 Starting the Software & Login Dialog Box Double click on the Endosafe Nexus icon to start the application. Upon starting the software, the login dialog box will be presented. Enter user name and password. User names and accounts are established by operators with administrator privileges. Permissions and access to some program functions are assigned based on the operator's user profile. If the account password has expired, the user will be notified after login and the Change Password dialog box will be displayed.
108 3 Main Window Endosafe Nexus System Controls The main application window of Endosafe Nexus consists of 7 icons: New Assay Configuration Administration Report View Log Help Exit
109 3.1 New Assay Icon Clicking on the New Assay icon will take the user to the assay pre-configure window.
110 3.1.1 Multi-Cartridge System Connection (Endosafe -MCS ) A status bar showing the connection status of Endosafe -MCS will appear and the message Please wait, connecting to the Multi Cartridge System will be displayed. If an error message is displayed stating the connection was not successful, verify that the MCS reader is powered on and properly connected to the computer. Next, click on the checkbox to reconnect the Endosafe Nexus to the MCS. Once the MCS is successfully connected, the message The Multi-Cartridge System is successfully connected will be displayed.
111 3.1.2 Nimbus Robot Connection When the MCS is successfully connected, the Nexus will start establishing a connection to the robot. A blue dotted circle animation, indicating the robot machine initialization is in progress, will appear along with the message Please wait, connecting to Nimbus robot. Once Nexus successfully connects to the robot, a green switch will appear indicating the connection to the Nimbus Robot was successful.
112 3.1.3 Assay Configuration By clicking the checkbox for this section, the user is prompted to enter to the Assay Configuration information. Enter the values for Total samples added and Total cartridges added. Note: The total number of cartridges must be greater than or equal to the total number of samples added, and must not exceed Nature of Sample Water By default, the nature of sample selected is Water. When the default sample (Water) is selected, the endotoxin limit is set to 0.25 and the max dosage unit is set to ml. Note: The max dosage unit cannot be changed when water is selected as a sample Nature of Sample Other When Others is selected as the sample, the max dosage unit and the endotoxin limit can be modified by the user. Selecting Others as the sample also enables the Concentration Dilution field, which can be modified by the user Choose Cartridge Lot This section contains the cartridge lot and corresponding calibration numbers that were entered and saved in the Administration Maintain Cartridge Data section. Select the desired cartridge lot numbers for the test cycle. When a cartridge lot number is selected, the corresponding calibration code and the type of cartridge are displayed. See the Administration section, 3.3.1, for instructions on how to enter new Cartridge Data.
113 Note: If multiple cartridge lot numbers are being used, the cartridges individual lot numbers and calibration codes can be changed in the review barcode table, after the Shift & Scan process Shift and Scan Barcode Select the checkbox for the Shift and Scan Barcode section to initiate the sample scanning process. When the checkbox is selected, the following confirmation dialog box will appear.
114 Click No and the last scan will be imported into the review barcode table after selecting OK in the window below. Then the following message will appear: Click Yes and all existing or previously scanned data will be cleared, including any scanned data that has been modified by the user, and the system will rescan the samples. A blue dotted circle animation along with the message Please wait, Nimbus robot sample scan is in progress will appear, as in the picture below.
115 Once the scanning process is complete, the red switch will turn into a green switch Review Barcode Note: The shift and scan review barcode tab will only be visible for bar code installation type. When the Shift and Scan Barcode process is complete, Nexus will populate the columns of the table on the right side of the screen. These columns represent the items selected in the configuration page. The Sample Name and Sample Lot values can be edited. The entered values for Sample Name, Sample Lot/Barcode, Custom Dilution/Conc., Endotoxin Limit, Nimbus Dilution, Reagent Location, can be copied to the other rows by clicking and dragging downs the cell to be copied while holding SHIFT. Clicking on the refresh icon (green icon in the upper right) will refresh the page and clear out all the entered and scanned information. In this window, any information can be changed to customize the automation. Multiple cartridge lot numbers, calibration codes, dilutions, endotoxin limits, sample names, and units can be used in the same automation. Move the cursor to the desired cell, double click in the field and update the information (with the exception of the Nimbus Dilution field, where double clicking will bring up the Dilution Table chart, for selection).
116 Nexus will display this message box upon trying to start the automation, if any information has been changed in the configuration table by the user: The user must then click the refresh button in the top-right corner of the program to make the Configuration Table changes to all samples. The refresh button will only update the columns that were changed in the Configuration Table. (e.g. changing the Endotoxin limit to 0.50). Until the user refreshes the grid to populate the newly configured value, the user cannot start the automation. BEFORE AFTER
117 3.1.6 Product Area The products database is a collection of the products a particular lab may test, as well as any products maintained in the Endoscan-V database. This information is pulled from the product database of the Endoscan-V software. If product data needs to be created or modified, this is done through Endoscan-V software, so please refer to Endoscan-V instructions. Note: In order to access the Endoscan-V s database, both programs must be accessed through the same user. The products area of this window provides user access to the Product Database during the Shift and Scan Barcode process. Products can be dragged and dropped to the review barcode table by 1) selecting the sample rows in Nexus that the user wishes to change/update and then 2) dragging the desired product from the bottom of the screen into the review barcode table. Note: -To select an individual sample, click on a row, in the column shown above in the red circle. -To select several samples, hold the CTRL key and click in the column shown above in the red circle, for each sample. -To select a group of samples, hold the SHIFT key and click in the column shown above in the red circle for the beginning row and then the end row of the group of samples.
118 The selected samples will then have their sample name, reagent location, and nimbus dilution changed to align with the EndoscanV database product s name, diluent, and Conc/Dil, respectively.
119 3.1.7 Dilutions Nexus can automatically perform dilutions on samples, from a 1:2 ratio up to a 1:300 ratio (termed Nexus Dilution ). In addition, one can pre-dilute samples prior to adding them, and Nexus can factor that Dilution factor into the final calculation (termed Custom Dilution). It is also possible for Nexus to further dilute a Custom Dilution. If the user double clicks on a Nimbus Dilution field, the following pop-up will appear, and the user can pick the desired dilution for the Nimbus robot to perform. When a dilution is selected the default reagent location is also chosen.
120 The Reagent Location column in the Sample Display Grid is where the user can change the location from which the reagent is drawn. See Maintain Reagent Location for more information. Note: Most diluted samples require two of each pipette tip, 1000µL and 300µL, per diluted sample which means that one can run up to 48 dilutions per run. For 1:2 dilutions, the robot will only use 300µL pipette tips for all steps, limiting the total number of possible samples with 1:2 dilutions in a run to 24. For any dilutions higher that 1:250, they system will use up more than the available reagents. Note: Dilutions and regular samples can be tested during the same automation. Upon starting a dilution, Nimbus will pick up reagent and place it into the samples respective dilution tube. Then, Nexus will replace the pipette tip and take some of the sample from the sample rack and place it into the respective dilution tube. After this, Nexus will replace the pipette tip and begin the mixing process. The dilution is now ready and Nexus replaces the pipette tip in order to place the sample into the PTS cartridge. While waiting for samples to finish testing, Nexus will begin the background task of diluting more samples. Once everything is set up correctly, clicking on the Start Automation button will start the automation process Automation Process Clicking on the Start Automation button will start the automation process Assay Real Time Status This section shows the real time status of the assay and the Nimbus movements, as well as all of the scanned and user entered information about each sample. This information can also be found in
121 individual assay reports, and the.csv file(s) in the Automation Summary folder Assay Run Status The 3 colored tabs at the top of the page display the current status of the assay. The Yellow tab shows the total number of assays remaining. The Red tab shows the total number of aborted assays. The Green tab shows the total number of completed assay. The Nimbus Status section shows whether or not the robot is in motion. The Pause Automation button will pause the automation and allow the user to add more samples to process. Clicking on the Exit Automation button will stop all the assays.
122 Pause Functionality Clicking the Pause button at the top of the Automation screen will park the robot at the next idle point and allow the user to add additional samples to the automation After the new Shift and Scan is complete or canceled, the following window will appear in the gray area above, and new entries can be customized with names, lot numbers, and dilutions.
123 By clicking the Merge Samples and Close button, Nexus will confirm the availability of the cartridges for the additional scanned samples. Verify the total number of cartridges in the dispenser including the additional samples and click Yes. Then, the samples can be merged with the existing samples and the automation will resume again.
124 Nimbus Movements This Tab shows all the movements of the Nimbus, like Inserting cartridges, adding samples, and removing cartridges for all bays. Bay 1, Bay 2, Bay 3, Bay 4 and Bay 5 will give the status of each bay, such as inserting the cartridge, adding sample, performing assay and removing cartridge Preparing the dilution as a background task Nexus will instruct Nimbus to pre-dilute the samples in the dilution rack that requires dilution. These prediluted samples will be used as the actual samples to test. The term BG Task: along with sample number is displayed in the Nimbus Movements panel so that the user is aware of the robotic movements.
125 The system continues to pre-dilute the samples that requires dilution as long as the MCS is busy running assays.
126 Assay Real Time Status of the Bays This tab shows the running status of the assays. After an assay is completed, the data for the Endotoxin Value and Result columns will be displayed. While the assay is in progress, right clicking on an individual assay will show the abort button for that particular assay. Clicking on abort will show a dialog box asking the user to enter comments as the reason for aborting the assay.
127 Starting a new automation cycle After every completed cycle the following restart procedure need to be followed. Exit the Nexus Automation software completely Power down the Nimbus robot instrument and power on after 5 minutes Execute the Nexus software Continue the automation cycle
128 3.2 Configuration From the Main Window, click on the Configuration to modify the configuration settings of the application Plate File Path This option is available to all of the operators. The plate file path is used as the default directory path to load and save template and plate files. To find a directory path, click the Browse button to display the following Browse For Folder screen. Folders with sub-folders are shown with an arrow ( ). To show the sub-folders, click the arrow or double-click the folder. When the OK button is clicked, the full path of the highlighted folder will be shown in the plate file path field. Clicking the cancel button will return the operator to the options dialog box.
129 3.2.2 Summary Report File Path This option is available to all operators. The summary report file path is used as the default directory path to save the summary report file path. To find a directory path, click the Browse button to display the following Browse For Folder screen Export File Path This option to export files is available to all operators. The export file path selected is used as the default directory path to save the export file of plate file data. To find a directory path, click the Browse button to display the following Browse For Folder screen Configuration Template Endotoxin Limit The limit of endotoxin expressed in EU/(dosage unit). Dosage Unit The dosage units are available from a pull-down menu or can be typed in. Concentration/Dilution The concentration or dilution of the product. If the dosage units are in ml, the dilution is expressed as a ratio (for example, 1:10). Otherwise, the unit is a concentration. Nexus Dilution Enter the dilutions to be made by the Nimbus robot, expressed as a ration (1:10). These dilutions are in addition to the custom dilutions/concentrations. Calibration Code Each lot of cartridges will have a unique calibration code based on the type of cartridge being used. Cartridge Lot Shows the cartridge lot for the type of cartridge being used. The cartridge lot number changes based on the type of cartridge. Sample Lot Barcode of the sample. Sample Name The trade name of the sample. This name will appear on reports for this sample. Batch Name An identification for multiple items in a single batch Scanned Time Time sample barcode was scanned. Sample Number Number of samples in the deck. Reagent Location Show the location of where the reagent is drawn Export Files The area to the right of the Configuration Template allows the user to enter the laboratories name, choose whether to save the files individually or as one consolidated.csv file.
130 3.3 Administration The administration button has a drop down button, Maintain Cartridge Data and Maintain Reagents Maintain Cartridge Data This section allows the user to add, edit or delete the cartridge lot and calibration codes Add New Cartridge lot and Calibration Codes Enter the cartridge lot and calibration code in the text boxes and click on Add New. When the data is successfully added, the message Save data successfully will appear and the new cartridge lot and calibration code will be added to the list.
131 Edit Cartridge lot and Calibration Codes Once the Edit icon is clicked, the cartridge lot and calibration code text box will get populated with the data of the selected row. Edit the desired information and click on the Update button in order to save the changes made Delete Cartridge lot and Calibration Codes To delete a cartridge and calibration code click on the Delete icon of the row to be deleted Maintain Reagent Locations The following picture will appear allowing the user to change the number assigned to each reagent location. The default locations are shown below.
132 3.4 Report Assay Report The Report Setup dialog box allows the operator to enter the report title and select the assay files to be included in the cartridge assay report. The report can be previewed, printed and ed (if the operator's machine supports ). The cartridge reader report provides one page per assay.
133 3.4.2 Preview Button Select the preview button on the Report Setup dialog box to preview the report on the computer screen. The report preview allows the operator to view all pages of the report and gives the operator the option to print the report. Note: Users may experience a delay while the report is being generated.
134 When done viewing the report, click on the close box in the right-hand corner of the screen to close the report preview and return to the main window Report Navigation To move through the report while in the preview mode, use the navigation buttons provided at the top of the window Report Search Report search allows the viewer to search the report for a specific word. Click on the search button to display a dialog box that allows entry of the desired text. The focus of the report preview window will move to the location of the entry in the report Report Zoom Report zoom adjusts the size of the report as it appears on the monitor. To see more of the page, select a lower percentage from the pull-down menu or type a percentage in the box. To see less of the page, select a higher percentage from the pull-down menu or type a percentage Export File Button The export button allows the user to save plate files under a different file type, such as.pdf Print Button The print button prints the report directly to the default printer. Selecting the print button a print dialog box. Make any necessary changes and select OK to print. displays
135 Button The button s the report to the recipient(s) identified in the dialog box. Selecting the button brings up the dialog box. Enter the recipient's address in the To line. Enter the addresses for anyone who should receive a copy of the report in the CC box. Enter a subject description in the Subject line. Any notes or comments can be typed into the main text area. Press the send button to send the and return to the main window or the cancel button to return to the main window Command Usage Log Report The command usage log stores information on commands used by operators. If command usage logging was enabled when Endosafe Nexus was installed, this menu command allows operators with administrator privileges to print the command usage report. The command usage report is in a PDF format Nimbus Report The Nimbus log stores all of the movements and functions of the Nimbus device. The Nimbus log allows the operators with administrator privileges to print the Nimbus Log. The Nimbus log report is in a PDF format.
136 3.5 View Log The view log has two sub tabs: Nimbus log Command usage log Nimbus Log The Nimbus log stores all the movements and functions of the Nimbus device and can be viewed in the by operators with administrator privileges.
137 3.5.2 Command Usage Log The command usage log stores information on all the commands done by operators and can be viewed by operators with administrator privileges. Note: The dialog box, the grid columns, and grid rows can be resized as needed.
138 Archive Clicking the Archive button presents a dialogue box to type a new file name. Once the file is saved, the currently displayed grid will be cleared and show the new log file.
139 3.6 Help The help section has two tabs: About Endosafe Nexus Displays the about screen, which provides software version and license information. Endosafe Nexus Manual Clicking on this will give the user the Endosafe Nexus manual. 3.7 Exit The Exit button will stop the Nexus software.
140 Instruções de Uso do Produto 4: Endosafe Microtrend (Software)
141 2008 Charles River
142 2 Endosafe MicroTrend Table of Contents Part I Endosafe MicroTrend 1 SP Quick Start Starting the Software Main Window Menus File Tools Help 5 Importing EndoScan-V PTS BioTrend Database Auto-Importing Data Table (Grid) Graphs Select Data Built-in Graphs Custom Graphs Reports Print Preview Print Save Report Audit Trail /... History Database Settings Search Part II Technical Support Charles River
143 Endosafe MicroTrend 1 SP Endosafe MicroTrend 1 SP 2 Endosafe MicroTrend provides a user interface to an assay database. The primary feature of the user interface is to graph the data to allow the user to visualize trends. 1.1 Quick Start A simple sequence of actions to familiarize with the capabilities of the EndoScan MicroTrend Finder software is: Register the software. Connect to a database (already accomplished using DBselector.exe). Import assays using one of the options in the File menu. Select a graph type from the pull down list Graph:. Select a date range to graph. Click Fill Lists and Data Table if the default dates were changed. Select an item from the first box. Select an item from the second box. Click Draw Graph. The divider between the grid and the graph areas can be moved to increase or decrease the desired viewing area. The double up arrow in the top right corner of the Select Data area can be clicked to alternately minimize or maximize the Select Data area. Report may be selected under the File menu to preview, print or save the graph information Charles River
144 4 Endosafe MicroTrend 2008 Charles River
145 Endosafe MicroTrend 1 SP Starting the Software Install License Screen Upon starting the software for the first time, an Install License screen is displayed. Fill in the Licensed To and License Key fields (License Key available from Charles River Labs - see Technical Support for contact information). After the correct information has been entered this screen will not be shown again Charles River
146 6 Endosafe MicroTrend Logo Screen Upon starting the software, a logo screen is displayed for approximately 10 seconds. The screen gradually becomes more solid. Following display of the logo screen the Main window will be presented. Note: The words "Special Edition" are shown when the custom version of the Endosafe MicroTrend program is used. This version can create custom graphs Charles River
147 Endosafe MicroTrend 1 SP Main Window The Main Window consists of the Menu Bar and three primary areas (or windows): the Select Data Area, the Grid Area and the Graph Area. Each window within the main window can be resized based on operator preference. Menu Bar The Menu Bar allows access to the following general menus. Each menu available from the Menu Bar is described in detail in their own help sections. File Menu The File Menu provides selection of various file formats to input to the MicroTrend database and options relating to reporting. Specific options include Import EndoScan csv and txt assays, Import PTS printout results and Import Biotrend database assays. Report and Exit are also available selections Charles River
148 8 Endosafe MicroTrend Tools Menu This menu identifies the tools available to the operator. Selections consist of Graph Settings, Import Settings, Database Search, and Database Settings. Help Menu The Help Menu provides access to online help available to the operator. The About menu item shows software version information and contact information for Charles River. 1.4 Menus File The Menu Bar allows access to the following general menus. Each menu available from the Menu Bar is described in detail in their own help sections. The File Menu provides the options for data filing and output. Specific options include Importing, Report and Exit. Import EndoScan-V Select an EndoScan export file (.csv or.txt). Import the assays in the export file to the MicroTrend database. Import PTS Select a PTS assay results file (.txt or.log). Import the assays in the assay results file to the MicroTrend database. Import Biotrend Select a Biotrend database file (.mdb). Import the assays in the export file to the MicroTrend database. Report Creates a report showing the selected parameters. Allows Report Preview, Printing and Saving a report to a file.. Exit Exit closes any open files and exits the operator from the program. Alternatively, clicking on the close box program. in the upper right-hand corner of the Main Window will also exit the 2008 Charles River
149 Endosafe MicroTrend 1 SP Tools This menu identifies the tools available to the operator and administrative options available (based on user privileges.) The tools available to the operator are Graph Settings, Import Settings, Database Search, and Database Settings. Graph Settings Graph properties may be set or changed including: the title, setting scaling ranges for the axis or setting to auto-scale and defining horizontal zones on the graph and selecting a color for each. Import Settings For EndoScan Text files, the Assay Date format can be set to Day Month Year, Month Day Year or Year Month Day. For PTS Log files, the Assay Date format can be set to Month Day Year or Day Month Year. These settings are also used by the AutoImport Service. Database Search Any column in the grid representation of the MicroTrend database may be searched for any character. Wild cards allow the desired character to be in any position in the string. Database Settings 2008 Charles River
150 10 Endosafe MicroTrend Database Settings displays the current server and database in use for the Endosafe MicroTrend database. In addition, a different Endosafe MicroTrend database may be selected or a new database created Help The Help Menu provides access to features to assist the operator in using the Endosafe MicroTrend program. The About Menu item provides software version, licensing and technical support information. Help The following screens and menus are available from the Help Menu located on the Menu Bar. Contents Contents displays help information organized by topic. Select the help topic by double-clicking on the book icon or clicking on the + to the left of the icon in the contents window to display the help topics. Clicking on a book or page icon will display the help information for that topic on the right side of the screen. Index Index displays help information by keyword. Enter the keywords for the desired help. A list of close matches will appear in the below the search box. Highlight the help topic in the lower window and select the Display button. The help information will be displayed on the right-hand side of the window Charles River
151 Endosafe MicroTrend 1 SP 2 11 Search Search displays help information after executing a search based on word(s) entered by the user. Enter the word(s) to search on and press the List Topics button. A list of previous searches can be accessed using the pull-down list on the text box. Topics containing the word or words are displayed in the Select Topic list box. To view a topic, highlight the topic title and press the display button. The help topic is displayed with the word or words highlighted. To turn off the highlighting, click the Options button on the toolbar, click the "Search Highlight Off" option, and click the Refresh button on the toolbar to start using the option. About Displays the About screen which provides software version and license information. The About screen also provides Technical Support contact information and a clickable link for comments. Note: The words "Special Edition" are shown when the custom version of the Endosafe MicroTrend Finder program is used. This version can create custom graphs Charles River
152 12 Endosafe MicroTrend 1.5 Importing Assay results can be imported manually using the EndoScan MicroTrend menu items or automatically. Endosafe MicroTrend imports files in four different formats: EndoScan-V version export.txt files, EndoScan-V version 3 and export.csv files, PTS printout files, Biotrend Jet database files. Note: Make sure the proper date format is set before auto-importing EndoScan.txt and PTS files. The date format may be changed using the Import Settings menu item in the Tools menu in the Endosafe MicroTrend program EndoScan-V 2008 Charles River
153 Endosafe MicroTrend 1 SP PTS 2008 Charles River
154 14 Endosafe MicroTrend BioTrend Database Auto-Importing Auto Importing is started automatically when Endosafe MicroTrend is installed or the computer is started. The subfolder Inbox is examined every 5 minutes, approximately. Any EndoScan export file, PTS printout file or Biotrend database file will be examined for assays not already in the MicroTrend database. After MicroTrend has processed the file it will be moved to the Outbox folder. A separate archive file, AAT, is kept for automatic import status Charles River
155 Endosafe MicroTrend 1 SP Data Table (Grid) The grid is populated by importing assays from EndoScan-V export files, PTS Printout files and Biotrend database files. The column order can be changed by dragging and dropping the column header in the new location. The column under the drop and all other columns to the right will be moved to the right. The +/- boxes can be used to expand the Standard Sets, Groups and Replicates 2008 Charles River
156 16 Endosafe MicroTrend 1.7 Graphs 2008 Charles River
157 Endosafe MicroTrend 1 SP Select Data Data to be graphed is selected from this screen. A number of built-in graphs are listed in the Graph drop down box. Select the desired graph type in the Graph drop down list box. Change the date range to be graphed as needed. When the graph type and/or date range is changed, the "Fill Lists and Data Table" button is enabled; click the button to update the graph data lists and the Data Table. The data group list box is loaded with the data groups in the database for the type of graph and selected date range. Click a data group to select it. Use the Shift or Ctrl keys with the mouse button to select multiple groups. The standard curve list box is loaded with the standard curves in the database for the selected date range. Click a standard curve to select it. Click the "Draw Graph" button to draw the graph Charles River
158 18 Endosafe MicroTrend Built-in Graphs The Graph drop down box includes the following built-in graphs: R by Analyst R by Reagent Y-intercept by Analyst Standard Optical Density by Analyst Standard Optical Density by Reagent Standard Reaction Time by Analyst Standard Reaction Time by Reagent Standard CVs by Analyst Product Data By Product Name Product Data by Product Type Product Data by Product Lot R by Accessory. The following graph shows R by Analyst for a single analyst Charles River
159 Endosafe MicroTrend 1 SP Custom Graphs Loading Custom Graphs To load a custom graph definition, click the "Load Custom Graph" button on the Select Data pane. Select the custom graph file and click the "Open" button. The graph definition will be added to the graph list on the Select Data pane. Using Custom Graphs Once a custom graph is loaded, it can be selected and used the same as built-in graphs Charles River
160 20 Endosafe MicroTrend 1.8 Reports The Report menu item allows Previewing, Printing and Saving of a graph. The Report Title can be set by the user Print Preview The Report Print Preview screen shows each of the pages of the report. Icons at the top of the screen allow a wide range of actions before printing. Actions include full, partial or multiple page display, magnifying an area and moving around the page, selecting the page or pages to view, searching for text in the report, printer setup and printing Charles River
161 2008 Charles River Endosafe MicroTrend 1 SP 2 21
162 22 Endosafe MicroTrend Print Printing is initiated from the Report Setup window or the Print Preview screen. A standard Windows print dialog box is used Charles River
163 Endosafe MicroTrend 1 SP Save Report The report is saved in PDF format to the folder and file name specified by the user Charles River
164 24 Endosafe MicroTrend 1.9 Audit Trail / History Two audit trails are created by MicroTrend. One when auto-importing, the other when the user performs certain actions. Actions Logged Actions logged include assay import, report printed or saved and audit trail archived. Successful imports are logged when auto-importing. Actions generating warnings and errors are not logged Database An existing SQL Server or MSDE MicroTrend database can be used or a new database can be created. Various graphs can be created from the data. Searches can be made on any column Settings The Database Utility creates Endosafe MicroTrend databases and saves database settings for MicroTrend. The Database Selector can be accessed directly from the folder that Endosafe MicroTrend is installed in (DBSelector.exe) or from the Endosafe MicroTrend program Tools menu, click Database Settings. Connection can be made to an existing SQL Server or MSDE database or a new database can be created Charles River
165 Endosafe MicroTrend 1 SP 2 25 The current database selection is shown in the "Settings" box. The settings are tested when the window is opened; any errors are displayed in the Errors box. To create a new database, enter the SQL Server 2000 or MSDE server name in the Server box. Use the server name or "(local)" for a server on the same PC as this utility. If the server has more than one instance, enter the instance name after the server name, e.g. "(local)\thirdinstance". Enter the new database name in the Database box; for example, you might use "MicroTrend". Click the "Create New Database" button to create the database. Note: The server must be running. To have MicroTrend use an existing database, enter the SQL Server 2000 or MSDE server name in the Server box. Use the server name or "(local)" for a server on the same PC as this utility. If the server has more than one instance, enter the instance name after the server name, e.g. "(local)\thirdinstance". Enter the database name in the Database box. Click the "Use Different Existing Database" button to change the settings. Note: The server must be running. You can close the window using the "Close" button or the button. Pressing the F1 key shows this help file Charles River
166 26 Endosafe MicroTrend Search Any column in the grid representation of the MicroTrend database may be searched for any character. Wild cards allow the desired character to be in any position in the string. Rows containing Standard Sets, Groups or Replicates can be expanded or collapsed using the buttons at the top of the screen Charles River
167 Endosafe MicroTrend 1 SP Technical Support For technical support contact: Charles River 1023 Wappoo Road, Suite 43B Charleston, SC Phone (843) Fax (843) [email protected] 2008 Charles River
168 Instruções de Uso do Produto 5: EndoScan-V version 5 (Software)
169
170
171
172
173
174
175
176
177
178
179
180
181
182
183
184
185
186
187
188
189
190
191
192
193
194
195
196
197
198
199
200
201
202
203
204
205
206
207
208
209
210
211
212
213
214
215
216
217
218
219
220
221
222
223
224
225
226
227
228
229
230
231
232
233
234
235
236
237
238
239
240
241
242
243
244
245
246
247
248
249
250
251
252
253
254
255
256
257
258
259
260
261
262
263
264
265
266
267
268
269
270
271
272
273
274
275
276
277
278
279
280
281
282
283
284
285
286
287
288
289
290
291
292
293
294
295
296
297
298
299
300
VGM. VGM information. ALIANÇA VGM WEB PORTAL USER GUIDE June 2016
 Overview The Aliança VGM Web portal is an application that enables you to submit VGM information directly to Aliança via our e-portal Web page. You can choose to enter VGM information directly, or to download
Overview The Aliança VGM Web portal is an application that enables you to submit VGM information directly to Aliança via our e-portal Web page. You can choose to enter VGM information directly, or to download
User Manual. Linksys PAP2 Broadband Phone Service. Linhagratuita grupo csdata
 User Manual Linksys PAP2 Broadband Phone Service Linhagratuita grupo csdata www.linhagratuita.com.br Please follow the step-by-step guide below to set up your Linksys PAP2 for use with Linhagratuita Broadband
User Manual Linksys PAP2 Broadband Phone Service Linhagratuita grupo csdata www.linhagratuita.com.br Please follow the step-by-step guide below to set up your Linksys PAP2 for use with Linhagratuita Broadband
LIGHT PANEL CONTENTS Light Panel Power supply Fixtures (brackets, screws)
 Page 1 of 8 LIGHT PANEL 20367 CONTENTS Light Panel Power supply Fixtures (brackets, screws) ABOUT THIS PRODUCT The Light Panel may be used on a table top as a free-standing object, or mounted either horizontally
Page 1 of 8 LIGHT PANEL 20367 CONTENTS Light Panel Power supply Fixtures (brackets, screws) ABOUT THIS PRODUCT The Light Panel may be used on a table top as a free-standing object, or mounted either horizontally
ATLAS DE ACUPUNTURA VETERINáRIA. CãES E GATOS (EM PORTUGUESE DO BRASIL) BY CHOO HYUNG KIM
 Read Online and Download Ebook ATLAS DE ACUPUNTURA VETERINáRIA. CãES E GATOS (EM PORTUGUESE DO BRASIL) BY CHOO HYUNG KIM DOWNLOAD EBOOK : ATLAS DE ACUPUNTURA VETERINáRIA. CãES E GATOS Click link bellow
Read Online and Download Ebook ATLAS DE ACUPUNTURA VETERINáRIA. CãES E GATOS (EM PORTUGUESE DO BRASIL) BY CHOO HYUNG KIM DOWNLOAD EBOOK : ATLAS DE ACUPUNTURA VETERINáRIA. CãES E GATOS Click link bellow
A necessidade da oração (Escola da Oração) (Portuguese Edition)
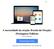 A necessidade da oração (Escola da Oração) (Portuguese Edition) Click here if your download doesn"t start automatically A necessidade da oração (Escola da Oração) (Portuguese Edition) A necessidade da
A necessidade da oração (Escola da Oração) (Portuguese Edition) Click here if your download doesn"t start automatically A necessidade da oração (Escola da Oração) (Portuguese Edition) A necessidade da
Introdução A Delphi Com Banco De Dados Firebird (Portuguese Edition)
 Introdução A Delphi Com Banco De Dados Firebird (Portuguese Edition) Ricardo De Moraes / André Luís De Souza Silva Click here if your download doesn"t start automatically Introdução A Delphi Com Banco
Introdução A Delphi Com Banco De Dados Firebird (Portuguese Edition) Ricardo De Moraes / André Luís De Souza Silva Click here if your download doesn"t start automatically Introdução A Delphi Com Banco
IDE 2.5" hd:basic. hdd enclosure caixa externa para disco rígido
 IDE 2.5" hd:basic hdd enclosure caixa externa para disco rígido IDE 2.5" hd:basic USER S GUIDE SPECIFICATONS HDD Support: IDE 2.5 Material: Aluminium Input connections: IDE HDD Output connections: USB
IDE 2.5" hd:basic hdd enclosure caixa externa para disco rígido IDE 2.5" hd:basic USER S GUIDE SPECIFICATONS HDD Support: IDE 2.5 Material: Aluminium Input connections: IDE HDD Output connections: USB
NORMAS DE FUNCIONAMENTO DOS CURSOS DE LÍNGUAS (TURMAS REGULARES E INTENSIVAS) 2015/2016
 NORMAS DE FUNCIONAMENTO DOS CURSOS DE LÍNGUAS (TURMAS REGULARES E INTENSIVAS) 2015/2016 1. Tipos de turma e duração: O CLECS oferece dois tipos de turma: regular e intensivo. Além destas turmas, o CLECS
NORMAS DE FUNCIONAMENTO DOS CURSOS DE LÍNGUAS (TURMAS REGULARES E INTENSIVAS) 2015/2016 1. Tipos de turma e duração: O CLECS oferece dois tipos de turma: regular e intensivo. Além destas turmas, o CLECS
Gestão da comunicação - Epistemologia e pesquisa teórica (Portuguese Edition)
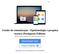 Gestão da comunicação - Epistemologia e pesquisa teórica (Portuguese Edition) Maria Cristina Castilho Costa, Maria Aparecida Baccega Click here if your download doesn"t start automatically Download and
Gestão da comunicação - Epistemologia e pesquisa teórica (Portuguese Edition) Maria Cristina Castilho Costa, Maria Aparecida Baccega Click here if your download doesn"t start automatically Download and
As 100 melhores piadas de todos os tempos (Portuguese Edition)
 As 100 melhores piadas de todos os tempos (Portuguese Edition) Click here if your download doesn"t start automatically As 100 melhores piadas de todos os tempos (Portuguese Edition) As 100 melhores piadas
As 100 melhores piadas de todos os tempos (Portuguese Edition) Click here if your download doesn"t start automatically As 100 melhores piadas de todos os tempos (Portuguese Edition) As 100 melhores piadas
1. Product Name. 2. Product Code. 3. Colour. 4. Brief Description. 5. Contents. 6. Snoezelen Stimulations. Switch Adapted Fibre Optic Lamp
 1. Product Name Switch Adapted Fibre Optic Lamp 2. Product Code 20709 3. Colour Clear base; white fibres 4. Brief Description Activate your switch to activate the light through the fibres and in the base.
1. Product Name Switch Adapted Fibre Optic Lamp 2. Product Code 20709 3. Colour Clear base; white fibres 4. Brief Description Activate your switch to activate the light through the fibres and in the base.
Um olhar que cura: Terapia das doenças espirituais (Portuguese Edition)
 Um olhar que cura: Terapia das doenças espirituais (Portuguese Edition) Padre Paulo Ricardo Click here if your download doesn"t start automatically Um olhar que cura: Terapia das doenças espirituais (Portuguese
Um olhar que cura: Terapia das doenças espirituais (Portuguese Edition) Padre Paulo Ricardo Click here if your download doesn"t start automatically Um olhar que cura: Terapia das doenças espirituais (Portuguese
FUTEBOL EXPLICA O BRASIL: UMA HISTóRIA DA MAIOR EXPRESSãO POPULAR DO PAíS, O (PORTUGUESE EDITION) BY MARCOS GUTERMAN
 Read Online and Download Ebook FUTEBOL EXPLICA O BRASIL: UMA HISTóRIA DA MAIOR EXPRESSãO POPULAR DO PAíS, O (PORTUGUESE EDITION) BY MARCOS GUTERMAN DOWNLOAD EBOOK : FUTEBOL EXPLICA O BRASIL: UMA HISTóRIA
Read Online and Download Ebook FUTEBOL EXPLICA O BRASIL: UMA HISTóRIA DA MAIOR EXPRESSãO POPULAR DO PAíS, O (PORTUGUESE EDITION) BY MARCOS GUTERMAN DOWNLOAD EBOOK : FUTEBOL EXPLICA O BRASIL: UMA HISTóRIA
Biscuit - potes (Coleção Artesanato) (Portuguese Edition)
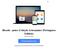 Biscuit - potes (Coleção Artesanato) (Portuguese Edition) Regina Panzoldo Click here if your download doesn"t start automatically Biscuit - potes (Coleção Artesanato) (Portuguese Edition) Regina Panzoldo
Biscuit - potes (Coleção Artesanato) (Portuguese Edition) Regina Panzoldo Click here if your download doesn"t start automatically Biscuit - potes (Coleção Artesanato) (Portuguese Edition) Regina Panzoldo
MySQL: Comece com o principal banco de dados open source do mercado (Portuguese Edition)
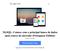 MySQL: Comece com o principal banco de dados open source do mercado (Portuguese Edition) Click here if your download doesn"t start automatically MySQL: Comece com o principal banco de dados open source
MySQL: Comece com o principal banco de dados open source do mercado (Portuguese Edition) Click here if your download doesn"t start automatically MySQL: Comece com o principal banco de dados open source
Medicina e Meditação - Um Médico Ensina a Meditar (Portuguese Edition)
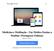 Medicina e Meditação - Um Médico Ensina a Meditar (Portuguese Edition) Roberto Cardoso Click here if your download doesn"t start automatically Medicina e Meditação - Um Médico Ensina a Meditar (Portuguese
Medicina e Meditação - Um Médico Ensina a Meditar (Portuguese Edition) Roberto Cardoso Click here if your download doesn"t start automatically Medicina e Meditação - Um Médico Ensina a Meditar (Portuguese
Pesquisa Qualitativa do Início ao Fim (Métodos de Pesquisa) (Portuguese Edition)
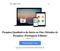 Pesquisa Qualitativa do Início ao Fim (Métodos de Pesquisa) (Portuguese Edition) Robert K. Yin Click here if your download doesn"t start automatically Pesquisa Qualitativa do Início ao Fim (Métodos de
Pesquisa Qualitativa do Início ao Fim (Métodos de Pesquisa) (Portuguese Edition) Robert K. Yin Click here if your download doesn"t start automatically Pesquisa Qualitativa do Início ao Fim (Métodos de
Livro do Desassossego
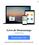 Livro do Desassossego Fernando Pessoa Click here if your download doesn"t start automatically Livro do Desassossego Fernando Pessoa Livro do Desassossego Fernando Pessoa [...] Download Livro do Desassossego...pdf
Livro do Desassossego Fernando Pessoa Click here if your download doesn"t start automatically Livro do Desassossego Fernando Pessoa Livro do Desassossego Fernando Pessoa [...] Download Livro do Desassossego...pdf
Computador móvel Dolphin TM Guia Rápido de Instalação
 Computador móvel Dolphin TM 6100 Guia Rápido de Instalação Computador móvel Dolphin 6100 Conteúdo da caixa Verifique se a caixa de seu produto contém os seguintes itens: Computador móvel Dolphin 6100
Computador móvel Dolphin TM 6100 Guia Rápido de Instalação Computador móvel Dolphin 6100 Conteúdo da caixa Verifique se a caixa de seu produto contém os seguintes itens: Computador móvel Dolphin 6100
Da Emoção à Lesão: um Guia de Medicina Psicossomática (Portuguese Edition)
 Da Emoção à Lesão: um Guia de Medicina Psicossomática (Portuguese Edition) Geraldo José Ballone, Ida Vani Ortolani, Eurico Pereira Neto Click here if your download doesn"t start automatically Download
Da Emoção à Lesão: um Guia de Medicina Psicossomática (Portuguese Edition) Geraldo José Ballone, Ida Vani Ortolani, Eurico Pereira Neto Click here if your download doesn"t start automatically Download
MANUAL DE MEDICINA INTERNA DE PEQUENOS ANIMAIS (EM PORTUGUESE DO BRASIL) BY RICHARD W. COUTO C. GUILHERMO^NELSON
 Read Online and Download Ebook MANUAL DE MEDICINA INTERNA DE PEQUENOS ANIMAIS (EM PORTUGUESE DO BRASIL) BY RICHARD W. COUTO C. GUILHERMO^NELSON DOWNLOAD EBOOK : MANUAL DE MEDICINA INTERNA DE PEQUENOS ANIMAIS
Read Online and Download Ebook MANUAL DE MEDICINA INTERNA DE PEQUENOS ANIMAIS (EM PORTUGUESE DO BRASIL) BY RICHARD W. COUTO C. GUILHERMO^NELSON DOWNLOAD EBOOK : MANUAL DE MEDICINA INTERNA DE PEQUENOS ANIMAIS
Como testar componentes eletrônicos - volume 1 (Portuguese Edition)
 Como testar componentes eletrônicos - volume 1 (Portuguese Edition) Renato Paiotti Newton C. Braga Click here if your download doesn"t start automatically Como testar componentes eletrônicos - volume 1
Como testar componentes eletrônicos - volume 1 (Portuguese Edition) Renato Paiotti Newton C. Braga Click here if your download doesn"t start automatically Como testar componentes eletrônicos - volume 1
National Institute for Space Research
 National Institute for Space Research Integrate and Testing Laboratory LIT TEST REPORT CUSTOMER: TEST ITEM: Desotec Comércio de Indústria de Equipamentos Eletrônicos Ltda Several MODEL: - REPORT NUMBER:
National Institute for Space Research Integrate and Testing Laboratory LIT TEST REPORT CUSTOMER: TEST ITEM: Desotec Comércio de Indústria de Equipamentos Eletrônicos Ltda Several MODEL: - REPORT NUMBER:
Editorial Review. Users Review
 Download and Read Free Online Java SE 8 Programmer I: O guia para sua certificação Oracle Certified Associate (Portuguese Edition) By Guilherme Silveira, Mário Amaral Editorial Review Users Review From
Download and Read Free Online Java SE 8 Programmer I: O guia para sua certificação Oracle Certified Associate (Portuguese Edition) By Guilherme Silveira, Mário Amaral Editorial Review Users Review From
Dermatologia Clínica. Guia Colorido Para Diagnostico e Tratamento (Em Portuguese do Brasil)
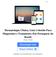 Dermatologia Clínica. Guia Colorido Para Diagnostico e Tratamento (Em Portuguese do Brasil) Click here if your download doesn"t start automatically Dermatologia Clínica. Guia Colorido Para Diagnostico
Dermatologia Clínica. Guia Colorido Para Diagnostico e Tratamento (Em Portuguese do Brasil) Click here if your download doesn"t start automatically Dermatologia Clínica. Guia Colorido Para Diagnostico
DIBELS TM. Portuguese Translations of Administration Directions
 DIBELS TM Portuguese Translations of Administration Directions Note: These translations can be used with students having limited English proficiency and who would be able to understand the DIBELS tasks
DIBELS TM Portuguese Translations of Administration Directions Note: These translations can be used with students having limited English proficiency and who would be able to understand the DIBELS tasks
Transformando Pessoas - Coaching, PNL e Simplicidade no processo de mudancas (Portuguese Edition)
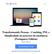 Transformando Pessoas - Coaching, PNL e Simplicidade no processo de mudancas (Portuguese Edition) Felippe / Marcelo Click here if your download doesn"t start automatically Transformando Pessoas - Coaching,
Transformando Pessoas - Coaching, PNL e Simplicidade no processo de mudancas (Portuguese Edition) Felippe / Marcelo Click here if your download doesn"t start automatically Transformando Pessoas - Coaching,
Manual dos locutores esportivos: Como narrar futebol e outros esportes no rádio e na televisão (Portuguese Edition)
 Manual dos locutores esportivos: Como narrar futebol e outros esportes no rádio e na televisão (Portuguese Edition) Carlos Fernando Schinner Click here if your download doesn"t start automatically Manual
Manual dos locutores esportivos: Como narrar futebol e outros esportes no rádio e na televisão (Portuguese Edition) Carlos Fernando Schinner Click here if your download doesn"t start automatically Manual
EXCEL PARA FINANçAS PESSOAIS (PORTUGUESE EDITION) BY FABRíCIO AUGUSTO FERRARI
 Read Online and Download Ebook EXCEL PARA FINANçAS PESSOAIS (PORTUGUESE EDITION) BY FABRíCIO AUGUSTO FERRARI DOWNLOAD EBOOK : EXCEL PARA FINANçAS PESSOAIS (PORTUGUESE EDITION) Click link bellow and free
Read Online and Download Ebook EXCEL PARA FINANçAS PESSOAIS (PORTUGUESE EDITION) BY FABRíCIO AUGUSTO FERRARI DOWNLOAD EBOOK : EXCEL PARA FINANçAS PESSOAIS (PORTUGUESE EDITION) Click link bellow and free
Estrelinhas Brasileiras: Como Ensinar a Tocar Piano de Modo Ludico Usando Pecas de Autores Brasileiros - Vol.2
 Estrelinhas Brasileiras: Como Ensinar a Tocar Piano de Modo Ludico Usando Pecas de Autores Brasileiros - Vol.2 Maria Ignes Scavone de Mello Teixeira Click here if your download doesn"t start automatically
Estrelinhas Brasileiras: Como Ensinar a Tocar Piano de Modo Ludico Usando Pecas de Autores Brasileiros - Vol.2 Maria Ignes Scavone de Mello Teixeira Click here if your download doesn"t start automatically
GUIÃO F. Grupo: Minho. 1º Momento. Intervenientes e Tempos. Descrição das actividades
 GUIÃO F Prova construída pelos formandos e validada pelo GAVE, 1/7 Grupo: Minho Disciplina: Inglês, Nível de Continuação 11.º ano Domínio de Referência: Um Mundo de Muitas Culturas 1º Momento Intervenientes
GUIÃO F Prova construída pelos formandos e validada pelo GAVE, 1/7 Grupo: Minho Disciplina: Inglês, Nível de Continuação 11.º ano Domínio de Referência: Um Mundo de Muitas Culturas 1º Momento Intervenientes
1. Product Name. 2. Product Code. 3. Colour. 4. Brief Description. 5. Contents. Peek A Boo Bear. Light brown bear the colour of the blanket may vary
 1. Product Name Peek A Boo Bear 2. Product Code 20837 3. Colour Light brown bear the colour of the blanket may vary 4. Brief Description Activate your switch to make this adorable bear play peek a boo.
1. Product Name Peek A Boo Bear 2. Product Code 20837 3. Colour Light brown bear the colour of the blanket may vary 4. Brief Description Activate your switch to make this adorable bear play peek a boo.
América Andina: integração regional, segurança e outros olhares (Portuguese Edition)
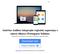 América Andina: integração regional, segurança e outros olhares (Portuguese Edition) Renata Peixoto de Oliveira, Silvia Garcia Nogueira, Filipe Reis Melo Click here if your download doesn"t start automatically
América Andina: integração regional, segurança e outros olhares (Portuguese Edition) Renata Peixoto de Oliveira, Silvia Garcia Nogueira, Filipe Reis Melo Click here if your download doesn"t start automatically
Como Falar no Rádio - Prática de Locução Am e Fm (Portuguese Edition)
 Como Falar no Rádio - Prática de Locução Am e Fm (Portuguese Edition) Cyro César Click here if your download doesn"t start automatically Como Falar no Rádio - Prática de Locução Am e Fm (Portuguese Edition)
Como Falar no Rádio - Prática de Locução Am e Fm (Portuguese Edition) Cyro César Click here if your download doesn"t start automatically Como Falar no Rádio - Prática de Locução Am e Fm (Portuguese Edition)
ATLAS DE ACUPUNTURA VETERINáRIA. CãES E GATOS (EM PORTUGUESE DO BRASIL) BY CHOO HYUNG KIM
 Read Online and Download Ebook ATLAS DE ACUPUNTURA VETERINáRIA. CãES E GATOS (EM PORTUGUESE DO BRASIL) BY CHOO HYUNG KIM DOWNLOAD EBOOK : ATLAS DE ACUPUNTURA VETERINáRIA. CãES E GATOS Click link bellow
Read Online and Download Ebook ATLAS DE ACUPUNTURA VETERINáRIA. CãES E GATOS (EM PORTUGUESE DO BRASIL) BY CHOO HYUNG KIM DOWNLOAD EBOOK : ATLAS DE ACUPUNTURA VETERINáRIA. CãES E GATOS Click link bellow
Quick user guide. (réf )
 Quick user guide (réf. 34889) Download the app «mobile eye door +» How to SETUP, for the first time, your Chacon IP VDP Connect the cables with the same color to each other, of both units. Connect the
Quick user guide (réf. 34889) Download the app «mobile eye door +» How to SETUP, for the first time, your Chacon IP VDP Connect the cables with the same color to each other, of both units. Connect the
O PRíNCIPE FELIZ E OUTRAS HISTóRIAS (EDIçãO BILíNGUE) (PORTUGUESE EDITION) BY OSCAR WILDE
 Read Online and Download Ebook O PRíNCIPE FELIZ E OUTRAS HISTóRIAS (EDIçãO BILíNGUE) (PORTUGUESE EDITION) BY OSCAR WILDE DOWNLOAD EBOOK : O PRíNCIPE FELIZ E OUTRAS HISTóRIAS (EDIçãO Click link bellow and
Read Online and Download Ebook O PRíNCIPE FELIZ E OUTRAS HISTóRIAS (EDIçãO BILíNGUE) (PORTUGUESE EDITION) BY OSCAR WILDE DOWNLOAD EBOOK : O PRíNCIPE FELIZ E OUTRAS HISTóRIAS (EDIçãO Click link bellow and
Pesquisa de Marketing: Uma Orientação Aplicada (Portuguese Edition)
 Pesquisa de Marketing: Uma Orientação Aplicada (Portuguese Edition) Naresh K. Malhotra Click here if your download doesn"t start automatically Pesquisa de Marketing: Uma Orientação Aplicada (Portuguese
Pesquisa de Marketing: Uma Orientação Aplicada (Portuguese Edition) Naresh K. Malhotra Click here if your download doesn"t start automatically Pesquisa de Marketing: Uma Orientação Aplicada (Portuguese
1. Product Name 2. Product Code 3. Colour 4. Brief Description 5. Contents 6. Snoezelen Stimulations
 1. Product Name Wheel Rotator 2. Product Code 13170, 16570, 20685 3. Colour Black 4. Brief Description A wheel rotator is needed to rotate the effect wheel in the ROMPA Projector. 5. Contents 1 x Wheel
1. Product Name Wheel Rotator 2. Product Code 13170, 16570, 20685 3. Colour Black 4. Brief Description A wheel rotator is needed to rotate the effect wheel in the ROMPA Projector. 5. Contents 1 x Wheel
Versão: 1.0. Segue abaixo, os passos para o processo de publicação de artigos que envolvem as etapas de Usuário/Autor. Figura 1 Creating new user.
 Órgão: Ministry of Science, Technology and Innovation Documento: Flow and interaction between users of the system for submitting files to the periodicals RJO - Brazilian Journal of Ornithology Responsável:
Órgão: Ministry of Science, Technology and Innovation Documento: Flow and interaction between users of the system for submitting files to the periodicals RJO - Brazilian Journal of Ornithology Responsável:
Guia de Instalação Rápida TWG-BRF114
 Guia de Instalação Rápida TWG-BRF114 Table Índice of Contents Português... 1. Antes de Iniciar... 2. Instalação de Hardware... 3. Configurando o Roteador... 1 1 2 3 Troubleshooting... 5 Version 09.24.2008
Guia de Instalação Rápida TWG-BRF114 Table Índice of Contents Português... 1. Antes de Iniciar... 2. Instalação de Hardware... 3. Configurando o Roteador... 1 1 2 3 Troubleshooting... 5 Version 09.24.2008
Bíblia do Obreiro - Almeida Revista e Atualizada: Concordância Dicionário Auxílios Cerimônias (Portuguese Edition)
 Bíblia do Obreiro - Almeida Revista e Atualizada: Concordância Dicionário Auxílios Cerimônias (Portuguese Edition) Sociedade Bíblica do Brasil Click here if your download doesn"t start automatically Bíblia
Bíblia do Obreiro - Almeida Revista e Atualizada: Concordância Dicionário Auxílios Cerimônias (Portuguese Edition) Sociedade Bíblica do Brasil Click here if your download doesn"t start automatically Bíblia
Planejamento de comunicação integrada (Portuguese Edition)
 Planejamento de comunicação integrada (Portuguese Edition) Click here if your download doesn"t start automatically Planejamento de comunicação integrada (Portuguese Edition) Planejamento de comunicação
Planejamento de comunicação integrada (Portuguese Edition) Click here if your download doesn"t start automatically Planejamento de comunicação integrada (Portuguese Edition) Planejamento de comunicação
Introdução A Delphi Com Banco De Dados Firebird (Portuguese Edition)
 Introdução A Delphi Com Banco De Dados Firebird (Portuguese Edition) Ricardo De Moraes / André Luís De Souza Silva Click here if your download doesn"t start automatically Introdução A Delphi Com Banco
Introdução A Delphi Com Banco De Dados Firebird (Portuguese Edition) Ricardo De Moraes / André Luís De Souza Silva Click here if your download doesn"t start automatically Introdução A Delphi Com Banco
Farmacologia na Pratica de Enfermagem (Em Portuguese do Brasil)
 Farmacologia na Pratica de Enfermagem (Em Portuguese do Brasil) Click here if your download doesn"t start automatically Farmacologia na Pratica de Enfermagem (Em Portuguese do Brasil) Farmacologia na Pratica
Farmacologia na Pratica de Enfermagem (Em Portuguese do Brasil) Click here if your download doesn"t start automatically Farmacologia na Pratica de Enfermagem (Em Portuguese do Brasil) Farmacologia na Pratica
ANATEL AGÊNCIA NACIONAL DE TELECOMUNICAÇÕES
 ANATEL AGÊNCIA NACIONAL DE TELECOMUNICAÇÕES GERÊNCIA DE CERTIFICAÇÃO E NUMERAÇÃO www.anatel.gov.br Visão geral do processo de certificação e homologação de produtos para telecomunicações no Brasil Lei
ANATEL AGÊNCIA NACIONAL DE TELECOMUNICAÇÕES GERÊNCIA DE CERTIFICAÇÃO E NUMERAÇÃO www.anatel.gov.br Visão geral do processo de certificação e homologação de produtos para telecomunicações no Brasil Lei
Manual do Usuário User Manual
 www.multilaser.com.br Manual do Usuário User Manual Índice/ Index ÍNDICE PORTUGUÊS Introdução...3 Descrição...3 Receptor...3 Transmissor...4 Configurando a Babá Eletrônica Baby Sound...4 1 Inserindo as
www.multilaser.com.br Manual do Usuário User Manual Índice/ Index ÍNDICE PORTUGUÊS Introdução...3 Descrição...3 Receptor...3 Transmissor...4 Configurando a Babá Eletrônica Baby Sound...4 1 Inserindo as
Atlas de Acupuntura Veterinária. Cães e Gatos (Em Portuguese do Brasil)
 Atlas de Acupuntura Veterinária. Cães e Gatos (Em Portuguese do Brasil) Atlas de Acupuntura Veterinária. Cães e Gatos (Em Portuguese do Brasil) O Atlas de Acupuntura Veterinária é um livro completo, em
Atlas de Acupuntura Veterinária. Cães e Gatos (Em Portuguese do Brasil) Atlas de Acupuntura Veterinária. Cães e Gatos (Em Portuguese do Brasil) O Atlas de Acupuntura Veterinária é um livro completo, em
Mitologia - Deuses, Heróis e Lendas (Portuguese Edition)
 Mitologia - Deuses, Heróis e Lendas (Portuguese Edition) By Maurício Horta, José Francisco Botelho, Salvador Nogueira Mitologia - Deuses, Heróis e Lendas (Portuguese Edition) By Maurício Horta, José Francisco
Mitologia - Deuses, Heróis e Lendas (Portuguese Edition) By Maurício Horta, José Francisco Botelho, Salvador Nogueira Mitologia - Deuses, Heróis e Lendas (Portuguese Edition) By Maurício Horta, José Francisco
GEAR PUMPS RECOMMENDATIONS BEFORE START-UP
 GEAR PUMPS RECOMMENDATIONS BEFORE START-UP CTI Gear pumps recommendations before start-up 0811-0 Attention: The application of ABER gear pumps must follow all the instructions hereby mentioned in order
GEAR PUMPS RECOMMENDATIONS BEFORE START-UP CTI Gear pumps recommendations before start-up 0811-0 Attention: The application of ABER gear pumps must follow all the instructions hereby mentioned in order
Biscuit - potes (Coleção Artesanato) (Portuguese Edition)
 Biscuit - potes (Coleção Artesanato) (Portuguese Edition) Regina Panzoldo Click here if your download doesn"t start automatically Biscuit - potes (Coleção Artesanato) (Portuguese Edition) Regina Panzoldo
Biscuit - potes (Coleção Artesanato) (Portuguese Edition) Regina Panzoldo Click here if your download doesn"t start automatically Biscuit - potes (Coleção Artesanato) (Portuguese Edition) Regina Panzoldo
GERENCIAMENTO PELAS DIRETRIZES (PORTUGUESE EDITION) BY VICENTE FALCONI
 Read Online and Download Ebook GERENCIAMENTO PELAS DIRETRIZES (PORTUGUESE EDITION) BY VICENTE FALCONI DOWNLOAD EBOOK : GERENCIAMENTO PELAS DIRETRIZES (PORTUGUESE Click link bellow and free register to
Read Online and Download Ebook GERENCIAMENTO PELAS DIRETRIZES (PORTUGUESE EDITION) BY VICENTE FALCONI DOWNLOAD EBOOK : GERENCIAMENTO PELAS DIRETRIZES (PORTUGUESE Click link bellow and free register to
Manual Instructions for SAP Note Version 1
 Manual Instructions for SAP Note 1973349 Version 1 TABLE OF CONTENTS 1 CHANGES TO CONTENT OF TABLE VIEW V_T596G... 3 2 CHANGES TO CONTENT OF TABLE VIEW V_T596I... 4 2 To perform the following changes,
Manual Instructions for SAP Note 1973349 Version 1 TABLE OF CONTENTS 1 CHANGES TO CONTENT OF TABLE VIEW V_T596G... 3 2 CHANGES TO CONTENT OF TABLE VIEW V_T596I... 4 2 To perform the following changes,
O sistema único de assistência social no Brasil: uma realidade em movimento (Portuguese Edition)
 O sistema único de assistência social no Brasil: uma realidade em movimento (Portuguese Edition) Click here if your download doesn"t start automatically O sistema único de assistência social no Brasil:
O sistema único de assistência social no Brasil: uma realidade em movimento (Portuguese Edition) Click here if your download doesn"t start automatically O sistema único de assistência social no Brasil:
Churrasco - Dando nome aos bois (Portuguese Edition)
 Churrasco - Dando nome aos bois (Portuguese Edition) István Wessel Click here if your download doesn"t start automatically Churrasco - Dando nome aos bois (Portuguese Edition) István Wessel Churrasco -
Churrasco - Dando nome aos bois (Portuguese Edition) István Wessel Click here if your download doesn"t start automatically Churrasco - Dando nome aos bois (Portuguese Edition) István Wessel Churrasco -
Minhas lembranças de Leminski (Portuguese Edition)
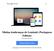 Minhas lembranças de Leminski (Portuguese Edition) Domingos Pellegrini Click here if your download doesn"t start automatically Minhas lembranças de Leminski (Portuguese Edition) Domingos Pellegrini Minhas
Minhas lembranças de Leminski (Portuguese Edition) Domingos Pellegrini Click here if your download doesn"t start automatically Minhas lembranças de Leminski (Portuguese Edition) Domingos Pellegrini Minhas
DO SILêNCIO DO LAR AO SILêNCIO ESCOLAR: RACISMO, PRECONCEITO E DISCRIMINAçãO NA EDUCAçãO INFANTIL (
 DO SILêNCIO DO LAR AO SILêNCIO ESCOLAR: RACISMO, PRECONCEITO E DISCRIMINAçãO NA EDUCAçãO INFANTIL ( DOWNLOAD EBOOK : DO SILêNCIO DO LAR AO SILêNCIO ESCOLAR: RACISMO, Click link bellow and free register
DO SILêNCIO DO LAR AO SILêNCIO ESCOLAR: RACISMO, PRECONCEITO E DISCRIMINAçãO NA EDUCAçãO INFANTIL ( DOWNLOAD EBOOK : DO SILêNCIO DO LAR AO SILêNCIO ESCOLAR: RACISMO, Click link bellow and free register
Poder sem limites - o caminho do sucesso pessoal pela programação neurolinguística
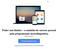 Poder sem limites - o caminho do sucesso pessoal pela programação neurolinguística Anthony Robbins Click here if your download doesn"t start automatically Poder sem limites - o caminho do sucesso pessoal
Poder sem limites - o caminho do sucesso pessoal pela programação neurolinguística Anthony Robbins Click here if your download doesn"t start automatically Poder sem limites - o caminho do sucesso pessoal
GERENCIAMENTO DA ROTINA DO TRABALHO DO DIA-A-DIA (EM PORTUGUESE DO BRASIL) BY VICENTE FALCONI
 Read Online and Download Ebook GERENCIAMENTO DA ROTINA DO TRABALHO DO DIA-A-DIA (EM PORTUGUESE DO BRASIL) BY VICENTE FALCONI DOWNLOAD EBOOK : GERENCIAMENTO DA ROTINA DO TRABALHO DO DIA-A- Click link bellow
Read Online and Download Ebook GERENCIAMENTO DA ROTINA DO TRABALHO DO DIA-A-DIA (EM PORTUGUESE DO BRASIL) BY VICENTE FALCONI DOWNLOAD EBOOK : GERENCIAMENTO DA ROTINA DO TRABALHO DO DIA-A- Click link bellow
1. Product Name. 2. Product Code. 3. Colour. 4. Brief Description. 5. Contents. Switch Adapted Clip On Fan. Colours may vary
 1. Product Name Switch Adapted Clip On Fan 2. Product Code 20489 3. Colour Colours may vary 4. Brief Description Connects to your switch for switch activation. 5. Contents Fan with cable with 3.5mm jack
1. Product Name Switch Adapted Clip On Fan 2. Product Code 20489 3. Colour Colours may vary 4. Brief Description Connects to your switch for switch activation. 5. Contents Fan with cable with 3.5mm jack
TDD Desenvolvimento Guiado por Testes (Portuguese Edition)
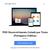 TDD Desenvolvimento Guiado por Testes (Portuguese Edition) Kent Beck Click here if your download doesn"t start automatically TDD Desenvolvimento Guiado por Testes (Portuguese Edition) Kent Beck TDD Desenvolvimento
TDD Desenvolvimento Guiado por Testes (Portuguese Edition) Kent Beck Click here if your download doesn"t start automatically TDD Desenvolvimento Guiado por Testes (Portuguese Edition) Kent Beck TDD Desenvolvimento
Ganhar Dinheiro Em Network Marketing (Portuguese Edition)
 Ganhar Dinheiro Em Network Marketing (Portuguese Edition) Click here if your download doesn"t start automatically Ganhar Dinheiro Em Network Marketing (Portuguese Edition) Ganhar Dinheiro Em Network Marketing
Ganhar Dinheiro Em Network Marketing (Portuguese Edition) Click here if your download doesn"t start automatically Ganhar Dinheiro Em Network Marketing (Portuguese Edition) Ganhar Dinheiro Em Network Marketing
O ABC da Fisioterapia Respiratória (Portuguese Edition)
 O ABC da Fisioterapia Respiratória (Portuguese Edition) George Jerre Vieira Sarmento (org.) Click here if your download doesn"t start automatically O ABC da Fisioterapia Respiratória (Portuguese Edition)
O ABC da Fisioterapia Respiratória (Portuguese Edition) George Jerre Vieira Sarmento (org.) Click here if your download doesn"t start automatically O ABC da Fisioterapia Respiratória (Portuguese Edition)
A ENTREVISTA COMPREENSIVA: UM GUIA PARA PESQUISA DE CAMPO (PORTUGUESE EDITION) BY JEAN-CLAUDE KAUFMANN
 Read Online and Download Ebook A ENTREVISTA COMPREENSIVA: UM GUIA PARA PESQUISA DE CAMPO (PORTUGUESE EDITION) BY JEAN-CLAUDE KAUFMANN DOWNLOAD EBOOK : A ENTREVISTA COMPREENSIVA: UM GUIA PARA CLAUDE KAUFMANN
Read Online and Download Ebook A ENTREVISTA COMPREENSIVA: UM GUIA PARA PESQUISA DE CAMPO (PORTUGUESE EDITION) BY JEAN-CLAUDE KAUFMANN DOWNLOAD EBOOK : A ENTREVISTA COMPREENSIVA: UM GUIA PARA CLAUDE KAUFMANN
Terras do Sem Fim (Em Portugues do Brasil)
 Terras do Sem Fim (Em Portugues do Brasil) Jorge Amado Click here if your download doesn"t start automatically Terras do Sem Fim (Em Portugues do Brasil) Jorge Amado Terras do Sem Fim (Em Portugues do
Terras do Sem Fim (Em Portugues do Brasil) Jorge Amado Click here if your download doesn"t start automatically Terras do Sem Fim (Em Portugues do Brasil) Jorge Amado Terras do Sem Fim (Em Portugues do
Guia de Instalação Rápida TE100-PCBUSR
 Guia de Instalação Rápida TE100-PCBUSR Table Índice of Contents Português... 1. Antes de Iniciar... 2. Como Instalar... 1 1 2 Troubleshooting... 3 Version 07.27.2007 1. Antes de Iniciar Conteúdo da Embalagem
Guia de Instalação Rápida TE100-PCBUSR Table Índice of Contents Português... 1. Antes de Iniciar... 2. Como Instalar... 1 1 2 Troubleshooting... 3 Version 07.27.2007 1. Antes de Iniciar Conteúdo da Embalagem
Direito Processual Civil (Coleção Sucesso Concursos Públicos e OAB) (Portuguese Edition)
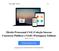 Direito Processual Civil (Coleção Sucesso Concursos Públicos e OAB) (Portuguese Edition) Marina Vezzoni Click here if your download doesn"t start automatically Direito Processual Civil (Coleção Sucesso
Direito Processual Civil (Coleção Sucesso Concursos Públicos e OAB) (Portuguese Edition) Marina Vezzoni Click here if your download doesn"t start automatically Direito Processual Civil (Coleção Sucesso
PL/SQL: Domine a linguagem do banco de dados Oracle (Portuguese Edition)
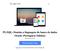 PL/SQL: Domine a linguagem do banco de dados Oracle (Portuguese Edition) Eduardo Gonçalves Click here if your download doesn"t start automatically PL/SQL: Domine a linguagem do banco de dados Oracle (Portuguese
PL/SQL: Domine a linguagem do banco de dados Oracle (Portuguese Edition) Eduardo Gonçalves Click here if your download doesn"t start automatically PL/SQL: Domine a linguagem do banco de dados Oracle (Portuguese
Vaporpunk - A fazenda-relógio (Portuguese Edition)
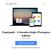 Vaporpunk - A fazenda-relógio (Portuguese Edition) Octavio Aragão Click here if your download doesn"t start automatically Vaporpunk - A fazenda-relógio (Portuguese Edition) Octavio Aragão Vaporpunk - A
Vaporpunk - A fazenda-relógio (Portuguese Edition) Octavio Aragão Click here if your download doesn"t start automatically Vaporpunk - A fazenda-relógio (Portuguese Edition) Octavio Aragão Vaporpunk - A
Gerenciamento Pelas Diretrizes (Portuguese Edition)
 Gerenciamento Pelas Diretrizes (Portuguese Edition) Vicente Falconi Click here if your download doesn"t start automatically Gerenciamento Pelas Diretrizes (Portuguese Edition) Vicente Falconi Gerenciamento
Gerenciamento Pelas Diretrizes (Portuguese Edition) Vicente Falconi Click here if your download doesn"t start automatically Gerenciamento Pelas Diretrizes (Portuguese Edition) Vicente Falconi Gerenciamento
BUTTONS & CONTROLS BOTÕES E CONTROLOS
 by BUTTONS & CONTROLS BOTÕES E CONTROLOS TALK* FALAR * PREVIOUS / NEXT* A NTERIOR / SE G U I NTE* POWER BUTTON & LED BOTÃO DE LIGAR E LED PAIRING BUTTON BOTÃO DE EMPARELHAMENTO LINE IN ENTRADA DE LINHA
by BUTTONS & CONTROLS BOTÕES E CONTROLOS TALK* FALAR * PREVIOUS / NEXT* A NTERIOR / SE G U I NTE* POWER BUTTON & LED BOTÃO DE LIGAR E LED PAIRING BUTTON BOTÃO DE EMPARELHAMENTO LINE IN ENTRADA DE LINHA
BUCHER COMERCIO DE EQUIPAMENTOS DE PESAGEM &TERCEIRIZAÇÃO DE SERVIÇOS EIRELI-ME Rua Izaura da S. Camargo, 39 Jardim São Paulo Cep: Taboão
 O calibrador Calog loadcell II é ideal para testes de 4 e 6 células de carga fio, verificando quebra do isolamento, pesando calibração do sistema e simulação. Pode testar uma célula de carga 'saldo zero,
O calibrador Calog loadcell II é ideal para testes de 4 e 6 células de carga fio, verificando quebra do isolamento, pesando calibração do sistema e simulação. Pode testar uma célula de carga 'saldo zero,
Erros que os Pregadores Devem Evitar (Portuguese Edition)
 Erros que os Pregadores Devem Evitar (Portuguese Edition) Ciro Sanches Zibordi Click here if your download doesn"t start automatically Erros que os Pregadores Devem Evitar (Portuguese Edition) Ciro Sanches
Erros que os Pregadores Devem Evitar (Portuguese Edition) Ciro Sanches Zibordi Click here if your download doesn"t start automatically Erros que os Pregadores Devem Evitar (Portuguese Edition) Ciro Sanches
Biossegurança: uma abordagem multidisciplinar (Portuguese Edition)
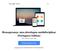 Biossegurança: uma abordagem multidisciplinar (Portuguese Edition) Pedro Teixeira, Silvio Valle Click here if your download doesn"t start automatically Biossegurança: uma abordagem multidisciplinar (Portuguese
Biossegurança: uma abordagem multidisciplinar (Portuguese Edition) Pedro Teixeira, Silvio Valle Click here if your download doesn"t start automatically Biossegurança: uma abordagem multidisciplinar (Portuguese
ISO/IEC 20000: Gerenciamento de Serviços de Tecnologia da Informação (Portuguese Edition)
 ISO/IEC 20000: Gerenciamento de Serviços de Tecnologia da Informação (Portuguese Edition) Jose Maria Fiorino Soula Click here if your download doesn"t start automatically ISO/IEC 20000: Gerenciamento de
ISO/IEC 20000: Gerenciamento de Serviços de Tecnologia da Informação (Portuguese Edition) Jose Maria Fiorino Soula Click here if your download doesn"t start automatically ISO/IEC 20000: Gerenciamento de
Dilogo Brasil Livro Texto: Curso Intensivo De Portugues Para Estrangeiros (Dialogo Brasil) (Portuguese Edition)
 Dilogo Brasil Livro Texto: Curso Intensivo De Portugues Para Estrangeiros (Dialogo Brasil) (Portuguese Edition) Emma Eberlein Lima, Samira Abirad Iunes, Marina Ribeiro Leite Click here if your download
Dilogo Brasil Livro Texto: Curso Intensivo De Portugues Para Estrangeiros (Dialogo Brasil) (Portuguese Edition) Emma Eberlein Lima, Samira Abirad Iunes, Marina Ribeiro Leite Click here if your download
HISTOLOGIA E BIOLOGIA CELULAR. UMA INTRODUçãO À PATOLOGIA (EM PORTUGUESE DO BRASIL) BY ABRAHAM L. KIERSZENBAUM
 Read Online and Download Ebook HISTOLOGIA E BIOLOGIA CELULAR. UMA INTRODUçãO À PATOLOGIA (EM PORTUGUESE DO BRASIL) BY ABRAHAM L. KIERSZENBAUM DOWNLOAD EBOOK : HISTOLOGIA E BIOLOGIA CELULAR. UMA INTRODUçãO
Read Online and Download Ebook HISTOLOGIA E BIOLOGIA CELULAR. UMA INTRODUçãO À PATOLOGIA (EM PORTUGUESE DO BRASIL) BY ABRAHAM L. KIERSZENBAUM DOWNLOAD EBOOK : HISTOLOGIA E BIOLOGIA CELULAR. UMA INTRODUçãO
Psicologia social contemporânea: Livro-texto (Portuguese Edition)
 Psicologia social contemporânea: Livro-texto (Portuguese Edition) Marlene Neves Strey, Pedrinho A. Guareschi, Maria da Graça Corrêa Jacques, Sérgio Antônio Carlos, Nara Maria G. Bernardes, Tania Mara Galli
Psicologia social contemporânea: Livro-texto (Portuguese Edition) Marlene Neves Strey, Pedrinho A. Guareschi, Maria da Graça Corrêa Jacques, Sérgio Antônio Carlos, Nara Maria G. Bernardes, Tania Mara Galli
Quick Setup Guide bluesound.com
 Quick Setup Guide bluesound.com Included Accessories 120V AC Power Cord 230V AC Power Cord Ethernet Cable Mini Jack to Toslink Adaptor 1 CONNECT ZO POWER 2 CONNECT TO WiFi A Select the AC Power Cord appropriate
Quick Setup Guide bluesound.com Included Accessories 120V AC Power Cord 230V AC Power Cord Ethernet Cable Mini Jack to Toslink Adaptor 1 CONNECT ZO POWER 2 CONNECT TO WiFi A Select the AC Power Cord appropriate
Transcript name: 1. Introduction to DB2 Express-C
 Transcript name: 1. Introduction to DB2 Express-C Transcript name: 1. Introduction to DB2 Express-C Welcome to the presentation Introduction to DB2 Express-C. In this presentation we answer 3 questions:
Transcript name: 1. Introduction to DB2 Express-C Transcript name: 1. Introduction to DB2 Express-C Welcome to the presentation Introduction to DB2 Express-C. In this presentation we answer 3 questions:
Direito Processual Civil (Coleção Sucesso Concursos Públicos e OAB) (Portuguese Edition)
 Direito Processual Civil (Coleção Sucesso Concursos Públicos e OAB) (Portuguese Edition) Marina Vezzoni Click here if your download doesn"t start automatically Direito Processual Civil (Coleção Sucesso
Direito Processual Civil (Coleção Sucesso Concursos Públicos e OAB) (Portuguese Edition) Marina Vezzoni Click here if your download doesn"t start automatically Direito Processual Civil (Coleção Sucesso
Bíblia do Obreiro - Almeida Revista e Atualizada: Concordância Dicionário Auxílios Cerimônias (Portuguese Edition)
 Bíblia do Obreiro - Almeida Revista e Atualizada: Concordância Dicionário Auxílios Cerimônias (Portuguese Edition) Sociedade Bíblica do Brasil Click here if your download doesn"t start automatically Bíblia
Bíblia do Obreiro - Almeida Revista e Atualizada: Concordância Dicionário Auxílios Cerimônias (Portuguese Edition) Sociedade Bíblica do Brasil Click here if your download doesn"t start automatically Bíblia
Adoção: guia prático doutrinário e processual com as alterações da Lei n , de 3/8/2009 (Portuguese Edition)
 Adoção: guia prático doutrinário e processual com as alterações da Lei n. 12010, de 3/8/2009 (Portuguese Edition) Luiz Antonio Miguel Ferreira Click here if your download doesn"t start automatically Adoção:
Adoção: guia prático doutrinário e processual com as alterações da Lei n. 12010, de 3/8/2009 (Portuguese Edition) Luiz Antonio Miguel Ferreira Click here if your download doesn"t start automatically Adoção:
Poder sem limites - o caminho do sucesso pessoal pela programação neurolinguística
 Poder sem limites - o caminho do sucesso pessoal pela programação neurolinguística Anthony Robbins Click here if your download doesn"t start automatically Poder sem limites - o caminho do sucesso pessoal
Poder sem limites - o caminho do sucesso pessoal pela programação neurolinguística Anthony Robbins Click here if your download doesn"t start automatically Poder sem limites - o caminho do sucesso pessoal
Completing your Participant Agreement Como preencher o Contrato de Participação
 Completing your Participant Agreement Como preencher o Contrato de Participação A quick-start guide for stock plan participants. Um guia rápido para participantes do plano de compra de ações. Your company
Completing your Participant Agreement Como preencher o Contrato de Participação A quick-start guide for stock plan participants. Um guia rápido para participantes do plano de compra de ações. Your company
Guia para Formacao de Analistas de Processos: Gestão Por Processos de Forma Simples (Portuguese Edition)
 Guia para Formacao de Analistas de Processos: Gestão Por Processos de Forma Simples (Portuguese Edition) Mr. Gart Capote Click here if your download doesn"t start automatically Guia para Formacao de Analistas
Guia para Formacao de Analistas de Processos: Gestão Por Processos de Forma Simples (Portuguese Edition) Mr. Gart Capote Click here if your download doesn"t start automatically Guia para Formacao de Analistas
Relações públicas comunitárias (Portuguese Edition)
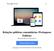 Relações públicas comunitárias (Portuguese Edition) Margarida Maria Krohling Kunsch Click here if your download doesn"t start automatically Relações públicas comunitárias (Portuguese Edition) Margarida
Relações públicas comunitárias (Portuguese Edition) Margarida Maria Krohling Kunsch Click here if your download doesn"t start automatically Relações públicas comunitárias (Portuguese Edition) Margarida
Addition of Fields in Line Item Display Report Output for TCode FBL1N/FBL5N
 Addition of Fields in Line Item Display Report Output for TCode FBL1N/FBL5N Applies to: Any business user who uses the transactions FBL1N and FBL5N to display line item reports for vendors and customers.
Addition of Fields in Line Item Display Report Output for TCode FBL1N/FBL5N Applies to: Any business user who uses the transactions FBL1N and FBL5N to display line item reports for vendors and customers.
Prática de Morfossintaxe: Como e por que Aprender Análise (Morfo)sintática (Portuguese Edition)
 Prática de Morfossintaxe: Como e por que Aprender Análise (Morfo)sintática (Portuguese Edition) Click here if your download doesn"t start automatically Prática de Morfossintaxe: Como e por que Aprender
Prática de Morfossintaxe: Como e por que Aprender Análise (Morfo)sintática (Portuguese Edition) Click here if your download doesn"t start automatically Prática de Morfossintaxe: Como e por que Aprender
Atividade Física Adaptada: Qualidade de Vida para Pessoas com Necessidades Especiais (Portuguese Edition)
 Atividade Física Adaptada: Qualidade de Vida para Pessoas com Necessidades Especiais (Portuguese Edition) Márcia Greguol Gorgatti, Roberto Fernandes da Costa Click here if your download doesn"t start automatically
Atividade Física Adaptada: Qualidade de Vida para Pessoas com Necessidades Especiais (Portuguese Edition) Márcia Greguol Gorgatti, Roberto Fernandes da Costa Click here if your download doesn"t start automatically
AAAASEL 0629 AAAA. Aplicação de Microprocessadores I. Prática 2 Teclado e Buzzer. Prof. Marcelo Andrade da Costa Vieira
 SEL 0629 Aplicação de Microprocessadores I Prática 2 Teclado e Buzzer Prof. Marcelo Andrade da Costa Vieira Teclado para tocar as 7 notas musicais no buzzer Objetivo: Aprendizado do microcontrolador PIC18F45K22
SEL 0629 Aplicação de Microprocessadores I Prática 2 Teclado e Buzzer Prof. Marcelo Andrade da Costa Vieira Teclado para tocar as 7 notas musicais no buzzer Objetivo: Aprendizado do microcontrolador PIC18F45K22
A ENTREVISTA COMPREENSIVA: UM GUIA PARA PESQUISA DE CAMPO (PORTUGUESE EDITION) BY JEAN-CLAUDE KAUFMANN
 Read Online and Download Ebook A ENTREVISTA COMPREENSIVA: UM GUIA PARA PESQUISA DE CAMPO (PORTUGUESE EDITION) BY JEAN-CLAUDE KAUFMANN DOWNLOAD EBOOK : A ENTREVISTA COMPREENSIVA: UM GUIA PARA CLAUDE KAUFMANN
Read Online and Download Ebook A ENTREVISTA COMPREENSIVA: UM GUIA PARA PESQUISA DE CAMPO (PORTUGUESE EDITION) BY JEAN-CLAUDE KAUFMANN DOWNLOAD EBOOK : A ENTREVISTA COMPREENSIVA: UM GUIA PARA CLAUDE KAUFMANN
Psicologia da Gratidão (Série Psicologica Joanna de Ângelis) (Portuguese Edition)
 Psicologia da Gratidão (Série Psicologica Joanna de Ângelis) (Portuguese Edition) Divaldo Franco, Joanna de Ângelis Click here if your download doesn"t start automatically Psicologia da Gratidão (Série
Psicologia da Gratidão (Série Psicologica Joanna de Ângelis) (Portuguese Edition) Divaldo Franco, Joanna de Ângelis Click here if your download doesn"t start automatically Psicologia da Gratidão (Série
Nome empresa: Criado por: Telefone:
 Texto da proposta Data: 22-1-216 SCALA2 3-45 A Grundfos SCALA2 is a fully integrated, self-priming, compact waterworks for pressure boosting in domestic applications. SCALA2 incorporates integrated speed
Texto da proposta Data: 22-1-216 SCALA2 3-45 A Grundfos SCALA2 is a fully integrated, self-priming, compact waterworks for pressure boosting in domestic applications. SCALA2 incorporates integrated speed
